��Ŀ����
��A��B��C��D���ֽ������ֱ�����Ǽ��뵽ϡ�����У�A��B��D��Ӧ��������ų���C�����������ٰ�A��D����B����Һ�У�D������B��������Aû�д����������ֽ����Ļ��˳��Ϊ( )
A. A��B��D��C B. B��D��A��C C. D��A��B��C D. D��B��A��C
D ���������������֪�����ڽ�A��B��C��D���ֽ������ֱ�Ͷ��ϡ�����У�C���ܷų����壬˵��C�Ľ�������������ٰ�A��DͶ��B����Һ�У�D������B������A��û�У�˵��D�Ļ�Ա�Bǿ��B�Ļ�Ա�Aǿ��������ֽ����Ļ��˳����D��B��A��C�����ԣ�D��ȷ��A��B��C����ѡD��ijУ��ѧ��ȤС�������ͭ��ͭ��п�Ͻ���ͭ��������������Ա��ȡ10g��ͭ��Ʒ�������з����ι�����30gϡ������Һ����ַ�Ӧ����ʵ�������£�
ʵ����� | 1 | 2 | 3 |
�������� | 10g | 10g | 10g |
�������� | 0.04g | m | 0.02g |
�Լ��㣺
��1���ڶ���ʵ���������������mΪ___________g��
��2����ͭ��ͭ��п�Ͻ��н���ͭ����������__________��
0.04g 67.5% ��������������ѧ֪ʶ��������Ϣ֪����1���ڶ���ʵ���������������mΪ0.04g����һ��ʵ���������������Ϊ0.04g��������ʵ���������������Ϊ0.02g��˵�����������в���п��û�з�Ӧ�ꡣ ��2�����������غ㶨��֪����������������0.04g ��0.04g ��0.02g��0.1g�� ������ͭ��п������Ϊx Zn��H2SO4��ZnSO4��H2�� ...ijͬѧ��һ��������ʯ��ʯ�м���7.3%��ϡ��������ȡ������̼����÷�Ӧ��������Һ��pH������ϡ����Ĺ�ϵ����ͼ��ʾ��
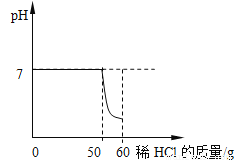
��1����ͬѧ���ռ����Ķ�����̼���������٣���д��������̣�
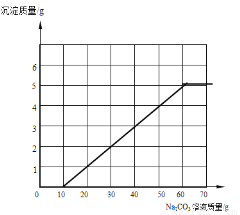
��2����ͬѧ����ȡ�����ķ�Һ���ˣ�����Һ�м���10.6%��Na2CO3��Һ����ü����Na2CO3��Һ������ij����������£�
Na2CO3��Һ����/g | 10 | 20 | 30 | 40 | 50 | 60 | 70 |
��������/g | 0 | 1 | n | 3 | 4 | 5 | m |
��m= ������10gNa2CO3��Һʱ������Ӧ�Ļ�ѧ����ʽ�� ��
������ͼ�л������Һ�м���Na2CO3��Һʱ�����Na2CO3��Һ������ij��������ߡ�