��Ŀ����
7��ijУ��ȤС���ͬѧȡij��ʯ��ʯ��Ʒ12g���вⶨʵ�飬�ֽ�100gϡ�������μ���ʯ��ʯ��Ʒ�У����ʲ�����ˮҲ�����뷴Ӧ������ַ�Ӧ������������������������ʾ��| ��1�� | ��2�� | ��3�� | ��4�� | ��5�� | |
| ����ϡ���������/g | 20 | 20 | 20 | 20 | 20 |
| ���������������/g | 1.1 | 2.2 | m | 4.4 | 4.4 |
��1��m��ֵΪ3.3g��
��2����4��Ϊ�պ���ȫ��Ӧ��
��3����ʯ��ʯ��Ʒ�Ĵ��ȣ�
���� ��1�����ݷ���μ��������ǰ���Σ�ÿ�ζ�����1.1g������̼�����ġ��嶼����4.4g������̼��˵��������û����ȫ��Ӧ��Ҳ����1.1g������mֵ��3.3g���з�����
��2�����ݵ��������ɶ�����̼������3.3g�����Ĵ����ɶ�����̼������4.4g�����ǵ���μ�������������̼�����������ӽ��з�����
��3������̼��ƺ����ᷴӦ�Ļ�ѧ����ʽ�����ɶ�����̼����������̼��Ƶ�������Ȼ�����̼��Ƶ�����������
��� �⣺��1������μ��������ǰ���Σ�ÿ�ζ�����1.1g������̼�����ġ��嶼����4.4g������̼��˵��������û����ȫ��Ӧ��Ҳ����1.1g������mֵ��3.3g��
��2�����������ɶ�����̼������3.3g�����Ĵ����ɶ�����̼������4.4g�����ǵ���μ�������������̼�����������ӣ����Ե�4��Ϊ�պ���ȫ��Ӧ��
��3����μӷ�Ӧ��̼�������Ϊx
CaCO3+2HCl=CaCl2+H2O+CO2��
100 44
x 4.4g
$\frac{100}{x}$=$\frac{44}{4.4g}$
x=10g
����ʯ��ʯ��Ʒ��̼��Ƶ���������Ϊ��$\frac{10g}{12g}$��100%=83.3%��
�ʴ�Ϊ����1��3.3��
��2��4��
��3��83.3%��
���� ������Ҫ�����˻�ѧ����ʽ�ļ��㣬�ѶȲ���ע�����Ĺ淶�ԣ�

 ��У����ϵ�д�
��У����ϵ�д�| A�� | �������⣻ú��ȼ�� | |
| B�� | ��ѩ�ڻ���ˮ������ʯ��ˮ��ͨ�������̼ | |
| C�� | ��ҩ��ը����ʯ���� | |
| D�� | ������ȼ����Һ�������������� |
 ��ͼ��ϡHCl��NaOH��Һ��Ӧ��pH�仯���ߣ�����˵����ȷ���ǣ�������
��ͼ��ϡHCl��NaOH��Һ��Ӧ��pH�仯���ߣ�����˵����ȷ���ǣ�������| A�� | �÷�Ӧ��ϡHCl����NaOH��Һ�� | |
| B�� | ag��ָ�����������ƹ������� | |
| C�� | B����Һ������ΪNaOH��NaCl | |
| D�� | ����A����Һ������Կ�����ɫ��̪��Һ |
| A�� | ���������������� | B�� | ϡ����������������� | ||
| C�� | �������ڼ��Ȳ��� | D�� | ���ʯ������̽������ͷ |
| A�� | ʯī�л���У��������缫 | B�� | �ɱ��������ȣ�������ʳƷ���ʼ� | ||
| C�� | ��̿�л�ԭ�ԣ������������� | D�� | ���ʯ��ɫ�����������и�� |

[����]��I������ȼ������CO��
������ȼ������CH4��
������ȼ������CO��CH4��
[�ռ�����]�ô�Ŀ�������ƿװ��ˮ����ƿ�������ӵ�����ˮ���У�����������Ȫˮƿ��ת��ʹƿ��ˮ����������ƿ�������ᵽ����Ѹ�ٸǺ�ƿ���ã�
[����ʵ��]
| ���� | ʵ������ | ʵ������ | ���� |
| ��1�� | ����ƿ�����壬ͨ��װ������������Һ��ϴ��ƿ�� | ���������� | Ŀ���dz�CO2���������壮 |
| ��2�� | ��ͨ��װ��Ũ�����ϴ��ƿ�� | ���������� | Ŀ���dz�ȥˮ�ݣ�ʹ������ |
| ��3�� | �����裨2�������嵼����ȼ�� ���ڻ����Ϸ���һ���ɶ����С�ձ� �� �� | ��ˮ����ͭ ����?�� ? | ?֤����H2O���ɣ� ?֤����CO2���ɣ� |
[��չ]����Ҫ���˽�����ˮ���������࣬����ˮ�����Ǻ�Ӧ���Ƚ��еIJ�����ͨ�磮
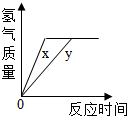 2015��5��12�գ��������ٹ�·������Ҫ��ɲ��֣�����ʡ��纣����--Ȫ���������ʽͨ�������ŵĽ��輰ͨ���漰������Ļ�ѧ֪ʶ��
2015��5��12�գ��������ٹ�·������Ҫ��ɲ��֣�����ʡ��纣����--Ȫ���������ʽͨ�������ŵĽ��輰ͨ���漰������Ļ�ѧ֪ʶ�� 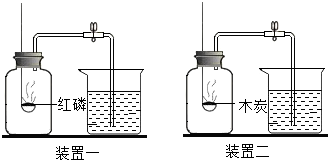 ��ͼ��ʾ�������ֲ�ͬ��ʵ��װ�ã�����ƿ�о�����������ȼ�ճ�����ʢ�е����� ��װ��һ������Ϊ���ף�װ�ö�������Ϊľ̿��װ�����зֱ�װ�к���ľ̿����Ϊ���������ձ���ʢ��ˮ�������ȼ�պ���ȴ�����£����ɼУ�
��ͼ��ʾ�������ֲ�ͬ��ʵ��װ�ã�����ƿ�о�����������ȼ�ճ�����ʢ�е����� ��װ��һ������Ϊ���ף�װ�ö�������Ϊľ̿��װ�����зֱ�װ�к���ľ̿����Ϊ���������ձ���ʢ��ˮ�������ȼ�պ���ȴ�����£����ɼУ�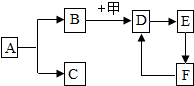 ��ͼ�е����ʾ��dz��л�ѧ�������ʣ�����A�dz�������ɫҺ�壬F�Ǵ���ʯ����Ҫ�ɷ֣����������ﳣ�õ�ȼ�ϣ���������֮���ת����ϵ����Щ��Ӧ��������Ӧ���������ʡ�ԣ����ش��������⣺
��ͼ�е����ʾ��dz��л�ѧ�������ʣ�����A�dz�������ɫҺ�壬F�Ǵ���ʯ����Ҫ�ɷ֣����������ﳣ�õ�ȼ�ϣ���������֮���ת����ϵ����Щ��Ӧ��������Ӧ���������ʡ�ԣ����ش��������⣺