��Ŀ����
�����д����л�ѧ��С����ѧ���Ļ�ѧ֪ʶ���������У���������̽����
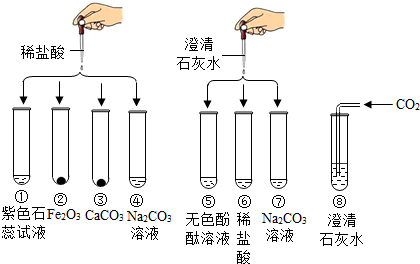
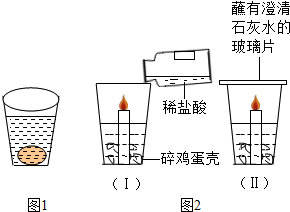
��1����ͼ1��ʾ��С����һֻ���ʼ�������ʢˮ�IJ������У���������ˮ�ף����ǵ���Ҫ�ɷ���̼��ƣ��������������μ�Ũ���ᣬ�����DZ��潫���ֵ������Ǽ����DZ��������ݲ�����Ӧ�Ļ�ѧ��Ӧ����ʽΪ ��
��2��С��ͬѧ�ڼ���������ͼ��ʾ��Сʵ�飮
��ͼ2�����й۲쵽����Ҫ�����Ǽ����DZ��������ݲ������������Ϩ���ɴ˵ó��й�CO2���ʵĽ����� �� ������������ȷ��CO2��һ����Ҫ��;�� ��
�ڵ�����Ϩ����ڲ�������Ѹ�ٸ�-��պ��ʯ��ˮ�IJ���Ƭͼ2�����۲쵽�������Dz���Ƭ�ϵ�ʯ��ˮ ��������˵���ڱ����� ���������д��������ʯ��ˮ��Ӧ�Ļ�ѧ����ʽ ��
��3������ˮ�೧�����ң�Ϊ�˲ⶨij��ɽʯ��ʯ��̼��Ƶ�����������ȡʯ��ʯ��Ʒ������ϡ�������ձ��з�Ӧ������ʯ��ʯ��Ʒ�����ʲ���ϡ���ᷴӦҲ������ˮ�����й�ʵ���������±���
���ʯ��ʯ��̼��Ƶ�����������
��1����ͼ1��ʾ��С����һֻ���ʼ�������ʢˮ�IJ������У���������ˮ�ף����ǵ���Ҫ�ɷ���̼��ƣ��������������μ�Ũ���ᣬ�����DZ��潫���ֵ������Ǽ����DZ��������ݲ�����Ӧ�Ļ�ѧ��Ӧ����ʽΪ

��2��С��ͬѧ�ڼ���������ͼ��ʾ��Сʵ�飮
��ͼ2�����й۲쵽����Ҫ�����Ǽ����DZ��������ݲ������������Ϩ���ɴ˵ó��й�CO2���ʵĽ�����
�ڵ�����Ϩ����ڲ�������Ѹ�ٸ�-��պ��ʯ��ˮ�IJ���Ƭͼ2�����۲쵽�������Dz���Ƭ�ϵ�ʯ��ˮ
��3������ˮ�೧�����ң�Ϊ�˲ⶨij��ɽʯ��ʯ��̼��Ƶ�����������ȡʯ��ʯ��Ʒ������ϡ�������ձ��з�Ӧ������ʯ��ʯ��Ʒ�����ʲ���ϡ���ᷴӦҲ������ˮ�����й�ʵ���������±���
| ��Ӧǰ | ��Ӧ�� | ||
| ʵ������ | �ձ���ϡ��������� | ʯ��ʯ��Ʒ������ | �ձ������л��������� |
| 160g | 12g | 167.6g | |
���㣺�εĻ�ѧ����,������̼�Ļ�ѧ����,���ݻ�ѧ��Ӧ����ʽ�ļ���
ר�⣺�йػ�ѧ����ʽ�ļ���,�������� ��ѧ����
��������1����������Ҫ�ɷ���̼��ƣ����������μ�Ũ���ᣬ��������Ӧ���ɶ�����̼���壬���ݷ�Ӧ��������д��ѧ����ʽ��
��2����������д�����ۺ���;�����������Ʋ���ۣ����ݷ�Ӧ��������д��ѧ����ʽ��
��3�����ݱ����е�������ͻ�ѧ����ʽ�����Լ����ʯ��ʯ��̼��Ƶ�����������
��2����������д�����ۺ���;�����������Ʋ���ۣ����ݷ�Ӧ��������д��ѧ����ʽ��
��3�����ݱ����е�������ͻ�ѧ����ʽ�����Լ����ʯ��ʯ��̼��Ƶ�����������
����⣺
��1�����ڵ�����Ҫ�ɷ���̼��ƣ����������μ�Ũ���ᣬ��������Ӧ���ɶ�����̼���壬������Ӧ�Ļ�ѧ����ʽΪ��CaCO3+2HCl�TCaCl2+H2O+CO2����
��2���ټ����ǵ���Ҫ�ɷ���̼��ƣ�̼�����ϡ���ᷴӦ�������ɶ�����̼���壬�������̼��֧��ȼ�գ���������ҲϨ���ˣ����Կ������ö�����̼��ȼ�գ�Ҳ��֧��ȼ�յ��ص������
�ڶ�����̼ʹ����ʯ��ˮ����ǣ����Բ���Ƭ�ϵ�ʯ��ˮ����ǣ����д�����������ʯ��ˮ��Ӧ�Ļ�ѧ����ʽΪ��CO2+Ca��OH��2�TCaCO3��+H2O��
��3�����ݱ����е����ݣ���֪������̼������Ϊ��160g+12g-167.6g=4.4g
�⣺��ʯ��ʯ��̼��Ƶ���������ΪX
CaCO3+2HCl�TCaCl2+CO2��+H2O
100 44
12gx 4.4g
��
=
�����x��83.3%
��ʯ��ʯ��̼��Ƶ���������Ϊ83.3%
�ʴ�Ϊ��
��1��CaCO3+2HCl�TCaCl2+H2O+CO2����
��2���ٶ�����̼��ȼ�ա�Ҳ��֧��ȼ�գ����
�ڱ���ǣ�������̼��CO2+Ca��OH��2�TCaCO3��+H2O��
��3��83.3%
��1�����ڵ�����Ҫ�ɷ���̼��ƣ����������μ�Ũ���ᣬ��������Ӧ���ɶ�����̼���壬������Ӧ�Ļ�ѧ����ʽΪ��CaCO3+2HCl�TCaCl2+H2O+CO2����
��2���ټ����ǵ���Ҫ�ɷ���̼��ƣ�̼�����ϡ���ᷴӦ�������ɶ�����̼���壬�������̼��֧��ȼ�գ���������ҲϨ���ˣ����Կ������ö�����̼��ȼ�գ�Ҳ��֧��ȼ�յ��ص������
�ڶ�����̼ʹ����ʯ��ˮ����ǣ����Բ���Ƭ�ϵ�ʯ��ˮ����ǣ����д�����������ʯ��ˮ��Ӧ�Ļ�ѧ����ʽΪ��CO2+Ca��OH��2�TCaCO3��+H2O��
��3�����ݱ����е����ݣ���֪������̼������Ϊ��160g+12g-167.6g=4.4g
�⣺��ʯ��ʯ��̼��Ƶ���������ΪX
CaCO3+2HCl�TCaCl2+CO2��+H2O
100 44
12gx 4.4g
��
| 100 |
| 12gx |
| 44 |
| 4.4g |
��ʯ��ʯ��̼��Ƶ���������Ϊ83.3%
�ʴ�Ϊ��
��1��CaCO3+2HCl�TCaCl2+H2O+CO2����
��2���ٶ�����̼��ȼ�ա�Ҳ��֧��ȼ�գ����
�ڱ���ǣ�������̼��CO2+Ca��OH��2�TCaCO3��+H2O��
��3��83.3%
�����������ѶȲ����ۺ��Խ�ǿ��������ͬѧ��������ѧ֪ʶ���з�����������������

��ϰ��ϵ�д�
�����Ŀ
����˵������ȷ���ǣ�������
| A���ճ�ʹ�õĽ������ϴ���Ǵ����� |
| B�����Ͻ��Ŵ���ɱ䰵��Ҫ��ɰֽ���˿����ĥ |
| C�����������Ԫ������Ȼ�����Ե�����ʽ���� |
| D��������Ǧ�ĵ�ػ����ˮ����������Ⱦ������Ӧ�����Ͼɵ�ص�ͳһ���մ��� |