��Ŀ����
��ѧ����������Ĺ�ϵԽ��Խ���С�
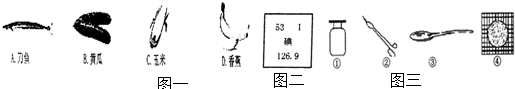
��1�����и���ʳ���е����ʺ�����ḻ���� _________������ţ���ͬ����
��1�����и���ʳ���е����ʺ�����ḻ���� _________������ţ���ͬ����

��2������ȱ����Ԫ���������״���״�Ԫ�������ڱ��е���Ϣ��ͼ��ʾ��������˵����ȷ����_________��
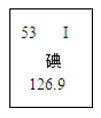
�ٵ�ʳ���еĵ���ָ�ⵥ�� �ڵ��ԭ��������53 �������߲ˡ��Ϲ��к��ḻ�ĵ�Ԫ�� �ܵ�Ԫ�ص����ԭ��������126.9
�ٵ�ʳ���еĵ���ָ�ⵥ�� �ڵ��ԭ��������53 �������߲ˡ��Ϲ��к��ḻ�ĵ�Ԫ�� �ܵ�Ԫ�ص����ԭ��������126.9
��3�����������е���Ʒ�����л��ϳɲ����Ƴɵ��� ____________ ��

��4�����Ǵ����·�ͨ��������ά֯�ɵģ���������ά��������ë�����ڵȣ������շ��ɳ���������������ά���������պ�����ս���ë��ζ����ά��____________��
���� ����ë �۵���
��5����ѧ��Ԥ�⡰���ܡ�����21���������������Դ������ȼ�յĻ�ѧ����ʽΪ_____________��
���� ����ë �۵���
��5����ѧ��Ԥ�⡰���ܡ�����21���������������Դ������ȼ�յĻ�ѧ����ʽΪ_____________��
��1����
��2���ڢ�
��3���٢�
��4����
��5��2H2 + O2 2H2O
2H2O
��2���ڢ�
��3���٢�
��4����
��5��2H2 + O2
 2H2O
2H2O 
��ϰ��ϵ�д�
 ͬ��������ϰϵ�д�
ͬ��������ϰϵ�д� �ο�ͨ�γ̱�˼ά����������ѵ��ϵ�д�
�ο�ͨ�γ̱�˼ά����������ѵ��ϵ�д�
�����Ŀ



 ��2009?��ƽ����ģ����ѧ����������Ĺ�ϵԽ��Խ���У�
��2009?��ƽ����ģ����ѧ����������Ĺ�ϵԽ��Խ���У�