��Ŀ����
ˮ������������ˮ��̫����������Ⱦ��ר���ڿ϶���������̫�����̵�ͬʱָ���������ֻ���α���Ҫ�α����뿿ȫ�������ӡ����д�ʩ��
�ٹ�����ҵ�ŷŵ�ˮҪ��ꣻ
�ڼӿ������ˮ����ϵͳ�Ľ��裻
�ۼ��ٻ��ʺ�ũҩ��ʹ������
���ᳫ��Լ��ˮ��
�ݽ��ú���ϴ�Ӽ���
�����ܸ���̫��ˮ�ʵ��ǣ� ����
A���٢ڢۢ�
B���ڢۢܢ�
C���٢ۢܢ�
D��ȫ��
D

��ϰ��ϵ�д�
 �㽭��У��ʦ���ϵ�д�
�㽭��У��ʦ���ϵ�д�
�����Ŀ
Ϊ��ȥ���������е����ʣ�������Ϊ���ʣ������в��������ܴﵽĿ�ĵ��ǣ� ��
| ѡ�� | ���ʣ����ʣ� |
|
| A | CO2��H2O�� | ������ͨ��ʢ�л��ĸ���� |
| B | Cu��CuO�� | ͨ���������������� |
| C | Na2SO4��Na2CO3�� | ��������ǡ�ò��ٲ�������Ϊֹ |
| D | FeCl2��CuCl2�� | ����������м����ַ�Ӧ����� |

 ����һʢ��100 mL 49��������Һ���ձ���ͬʱ��һ����⻬������С����������Һ���루��ͼ��ʾ�����������Һ�л���ע��20����Ba(OH)2��Һ��ǡ����ȫ��Ӧ���ڴ�ʵ������У�
����һʢ��100 mL 49��������Һ���ձ���ͬʱ��һ����⻬������С����������Һ���루��ͼ��ʾ�����������Һ�л���ע��20����Ba(OH)2��Һ��ǡ����ȫ��Ӧ���ڴ�ʵ������У� ����
���� ��������
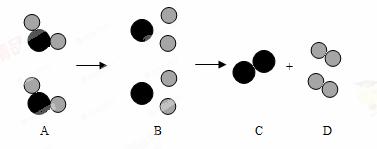
�������� ����ʾ��ԭ�ӣ���
����ʾ��ԭ�ӣ��� ����ʾ��ԭ�ӣ���
����ʾ��ԭ�ӣ��� ����ʾˮ
����ʾˮ ���Ӹ���ģ��ͼ
���Ӹ���ģ��ͼ �ش�
�ش�