��Ŀ����
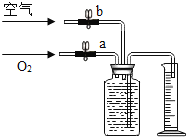
��ͼ1�ļ��ȴ�������Ұ�����ʳ����ȴ��еĹ����ĩ��þ�ۡ����ۺ��Ȼ��ƣ�ʹ��ʱ�����м���ˮ���е�ˮ�����ɲ��������ȡ�ʵ��С����Է��ȹ����еķ�Ӧԭ��չ��̽����
���������ϣ�
������þ������ˮ�����û���Ӧ���ҷ��ȡ�

������ʵ�飩
ͬѧ����ͼ2��ʾװ�ý���ģ��ʵ�飺�ֱ�ȡ��ͬ�ɷֵĹ����ĩ������У�ͨ����Һ©�������о�����8mLˮ����ȡ�������ù�Һ����������¶ȣ�ʵ���¼���±�����ʵ��ʱ������Ϊ22.8�棩
ʵ����� | A | B | C | D | E | F |
����ɷ� | Mg | Fe | Mg+Fe | Mg+NaCl | Fe+NaCl | Mg+Fe+NaCl |
�������� | ���������ݣ����Ե�ȼ | _____ | ���������ݣ����Ե�ȼ | �϶�����ݣ���ȼ�б����� | ������ | ���������ݣ���ȼ�б����� |
������¶� | 23.1�� | 22.8�� | 23.1�� | 24.2�� | 22.8�� | 27.2�� |
����������ۣ�
��1��ʵ��B����������Ϊ_____��
��2��ʵ��A֤����þ����ˮ�ܷ�Ӧ����ɸ÷�Ӧ�Ļ�ѧ����ʽMg+2H2O��_____+H2��
��3��ʹþ����ˮѸ�ٷ�Ӧ�����ȵ���ѷ�������þ���м���_____��
����˼�Ľ���
��4��ͬѧ�Ƿ���ʵ�����ݷ��֣����ߵ��¶�û�дﵽʳƷ���ȴ���Ч��������ܵ�ԭ����_____��
��5��ͬѧ�Ǹ���ʵ������һ���²⣬ʵ��F�еķ�Ӧ��������ʣ�������������Ȼ�������ۣ����������۴��ڵ�ʵ�鷽��Ϊ��ȡ������������_____��
��6��������ʵ������ó���NaCl����ˮû�������ı仯���Ľ��ۣ����ݵ�����ʵ���ǣ���ʵ����ţ�_____��
���й��ɺ��ܽ���ȫ��ȷ��һ���ǣ� ��
A����ѧ��Ӧ�������� | B����ѧ��Ӧ�г����ġ����� |
�ٻ��Ϸ�Ӧ��4P+5O2 �ڷֽⷴӦ��H2CO3�TH2O+CO2�� ���û���Ӧ��3CO+Fe2O3 | ��ú��ʯ�͡���Ȼ��������ʯȼ�� �ڿ�ȼ��������¶ȴﵽ�Ż�㣬ȼ�������� �۷��ӡ�ԭ�ӡ����ӣ��������ʵ��������� |
C���Լ�������ʶ | D����ѧ������ |
�����ֵ�������������������ǵ�ľ�� ����������ά����ë��ά����ȼ������ζ ����������狀��Ȼ�泥�����ʯ�ҷ�ĩ��ĥ | ���ü�ȩˮ��Һ����ˮ��Ʒ���� ��ȱ��ά����C����Ѫ�� �����г�֧����������� |
A. A B. B C. C D. D


 2P2O5
2P2O5 2Fe+3CO2
2Fe+3CO2