��Ŀ����
17����ͼ��ͬѧ�ǹ����Ĺ��������IJ���֪ʶ���磨����C6H12O6Ϊ�����ǣ�����ش�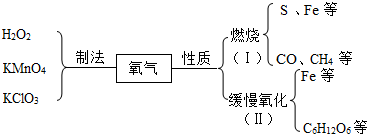
��1��ʵ�����ù�������ֽ���ȡ�����Ļ�ѧ����ʽ��2H2O2$\frac{\underline{\;MnO_2\;}}{\;}$2H2O+O2����
��2��ͼ�г��ֵ������У�����������Ļ�ѧʽ�У�дһ�֣�H2O2����CO����
I��II�����������μӵĻ�ѧ��Ӧ����ͬ���ǣ�дһ��������������Ӧ���ų���������
��3���������ɷ��ӣ�����ӡ�����ԭ�ӡ������ӡ����ɣ���
��4�����������������˵����������������Ĺ�ϵ����CH4ȼ�գ��ṩ�������������⣬�˷ѽ�����Դ��
���� ��1������ʵ�����ù�������ֽ���ȡ������д����Ӧ�Ļ�ѧ����ʽ���
��2������������Ķ��壬��ѧ��Ӧԭ�����
��3�������������������ӹ��ɵĽ��
��4����������������ף�
��� �⣺
��1��ʵ�����ù�������ֽ���ȡ�����Ļ�ѧ����ʽΪ��2H2O2$\frac{\underline{\;MnO_2\;}}{\;}$2H2O+O2��
��2����������������Ԫ����ɣ�����һ������Ԫ�صĻ��������������ͼ�г��ֵ������У�����������Ļ�ѧʽ�У�H2O2����CO��
���ʵ�ȼ�պͻ��������Ĺ�ͬ�㣺����������Ӧ���ų���������
��3���������������ӹ��ɣ�
��4���κ����ﶼ�������ԣ�������CH4ȼ�գ��ṩ���� ���������⣬�˷ѽ�����Դ���������ɣ���
�𰸣�
��1��2H2O2$\frac{\underline{\;MnO_2\;}}{\;}$2H2O+O2��
��2��H2O2����CO�� ����������Ӧ���ų�������
��3������
��4������CH4ȼ�գ��ṩ���� ���������⣬�˷ѽ�����Դ
���� ���⿼������������ȡ�����ʣ�������д��Ӧ�ķ���ʽ����ȷ������Ӧ�������ǽ���Ĺؼ����ѶȲ���

��ϰ��ϵ�д�
�����Ŀ
7��������ѧ����¶α��ѧ���ǿ�ѧ�����ߵ��������и���������α��ѧ���ǣ�������
| A�� | �øɱ������˹����� | B�� | ��ʯ���Զ��һ�������� | ||
| C�� | �ô�����ˮ������� | D�� | ��Һ̬�ⷢ���� |
8������Ͳ��ȡҺ��ʱ��ijͬѧ�����Ӱ�Һ�����ʹ�������Ϊ19mL���㵹һ����Һ�壬�ָ��Ӱ�Һ����ʹ�������Ϊ13mL������λͬѧȡ����Һ������ǣ�������
| A�� | ����6mL | B�� | С��6mL | C�� | ����6mL | D�� | ���ж� |
5�����л�ѧ��Ӧ�У������û���Ӧ���ǣ�������
| A�� | C+O2$\frac{\underline{\;��ȼ\;}}{\;}$CO2 | B�� | Zn+2HCl�TZnCl2+H2�� | ||
| C�� | CO+CuO$\frac{\underline{\;����\;}}{\;}$Cu+CO2 | D�� | CaCO3$\frac{\underline{\;����\;}}{\;}$CaO+CO2�� |
12����ͼ��ij��ѧ��Ӧ����ģ�ͣ���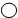 ������
������ ���ֱ��ʾ��ͬԪ�ص�ԭ�ӣ����ж�ͼʾģ��������ȷ���ǣ�������
���ֱ��ʾ��ͬԪ�ص�ԭ�ӣ����ж�ͼʾģ��������ȷ���ǣ�������
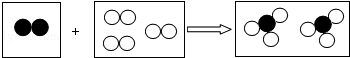
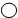 ������
������ ���ֱ��ʾ��ͬԪ�ص�ԭ�ӣ����ж�ͼʾģ��������ȷ���ǣ�������
���ֱ��ʾ��ͬԪ�ص�ԭ�ӣ����ж�ͼʾģ��������ȷ���ǣ�������
| A�� | �÷�Ӧ���ڷֽⷴӦ | |
| B�� | ��Ӧ�����������ֻ��һ�ֵ��� | |
| C�� | ��Ӧ�й�������Ԫ�ز��룬���������Ϊ������ | |
| D�� | �μӷ�Ӧ���������ʷ��Ӹ���֮��Ϊ2��3 |
9�����з����м��ܴ���һ��Ԫ�أ����ܴ���һ��ԭ�ӣ����ܴ���һ�����ʵ��ǣ�������
| A�� | Fe | B�� | O2 | C�� | H | D�� | 2H |
6�������������ŷɴ��õ��dz�����F���ͻ�����û����ȼ����ƫ������[��CH3��2N-NH2]����������������������N2O4���������й�˵����ȷ���ǣ�������
| A�� | ��������У�ʧ�������£��ȵ�������������ʵ�� | |
| B�� | ƫ��������̼ԭ�ӡ�����ӡ���������� | |
| C�� | ƫ��������2��̼ԭ�ӡ�8����ԭ�ӡ�2����ԭ�ӹ��� | |
| D�� | �������������Ԫ�ص�������������Ϊ1��2 |