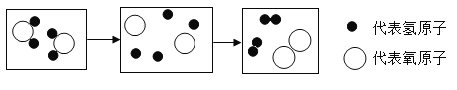
��Ŀ����
�����ᾧˮ�ļס������ֹ������ʵ��ܽ��������ͼ��ʾ������˵����ȷ����
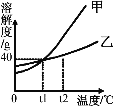
A. t1��ʱ���ס��ұ�����Һ�к����ʵ��������
B. t1��ʱ���ס����������ʸ�25g�ֱ���뵽50gˮ�У�����ܽ⣬�õ�����Һ��������70g
C. t2��ʱ������Һ��������������һ����������Һ��������������
D. t2��ʱ�ס��ҵı�����Һ���µ�t1��ʱ�����������������һ��������
С����������������Һ��Ӧ��ʵ��ʱ���������ɵ������д̼�����ζ�����ǽ�����̽����
��������⣩����������Һ��Ӧ���ɵ�����Ϊʲô�д̼�����ζ��
���������ϣ�
(1)6H2SO4(Ũ)+2Fe Fe2(SO4)3+6H2O+3SO2��
Fe2(SO4)3+6H2O+3SO2��
(2)SO2��ʹƷ����Һ�ĺ�ɫ��ȥ
�����в��룩���벻ͬŨ�ȵ�������Һ��Ӧ�����ɵ�������������ж�������
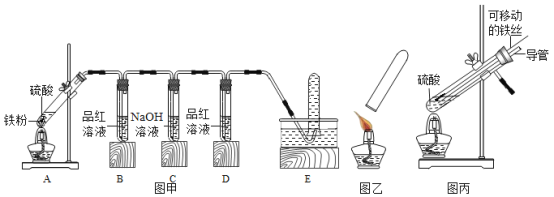
��ʵ��̽����С����ͼ����ʾװ�ý���ʵ�飬����E���ռ��������������ͼ����ʾ�ı���ʵ�飮
(1)�����С������±���
ʵ������ | A����������ɷ� | ||||
B��Ʒ����Һ | D��Ʒ����Һ | ����ʵ�� | |||
ʵ��һ | 98% | ________ | ����ɫ | �ޱ����� | ֻ��SO2 |
ʵ��� | 45% | ������ɫ | ����ɫ | �б����� | ________ |
ʵ���� | 25% | ________ | ����ɫ | ________ | ֻ��H2 |
��ʵ����ۣ����벻ͬŨ�ȵ����ᷴӦ�����ɵ����������ܲ�ͬ����������ҺŨ�ȴﵽ�㹻��ʱ�����ɵ�����������ж�������
(2)��ʵ����ۣ����벻ͬŨ�ȵ����ᷴӦ�����ɵ����������ܲ�ͬ����������ҺŨ�ȴﵽ�㹻��ʱ�����ɵ�����������ж�������
��������˼��
(1)д��ʵ����������������Һ��Ӧ�Ļ�ѧ����ʽ ________ ��д��ʵ����з���������Ӧ�Ļ�ѧ����ʽ________ ��
(2)ʵ��һ��Cװ�õ�������________ ��
����չ���죩С���ֽ�Aװ�ý�������ͼ��ʾ�ĸĽ��������ӵĵ����¶�����Һ������ԭ��________ ���õ��������������_________ ��


 �Ž��з� B.
�Ž��з� B.  �����
����� ������ D.
������ D.  ţ��
ţ��