��Ŀ����
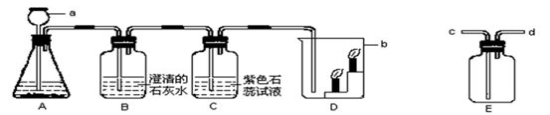
(6��)ʵ������һ�Ȼ��ƺ��Ȼ�����ɵ���Ʒ��Ϊ�ⶨ�����Ȼ��ƵĴ��Ȳ����Ƴ�һ������������������Һ��ij��ѧ̽��С��ȡһ�������ĸ���Ʒ��������ʵ�顣
��ش��������⣺
��1��д����Ӧ�ڵĻ�ѧ����ʽ ��
��2��������֪�����г������вμӷ�Ӧ�Ĵ�������(X)�ı���ʽ
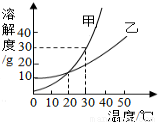
��3������Ʒ���Ȼ��Ƶ���������Ϊ ��
��4������36.5%��Ũ������������ǡ����ȫ��Ӧ��ϡ���ᣬ���ˮ�����Ϊ mL��
��5�����������õ�c��ҺΪֻ��һ�����ʵIJ�������Һ����c��Һ�����ʵ���������Ϊ ��
��6������ҵ����58.5t�������Ȼ��Ƶ���ƷΪԭ������ȡ�������ƣ����Ƶ��������Ƶ�����Ϊ ��
��1��Na2CO3+ 2HCl =2 NaCl+ CO2��+H2O����2�� =
= ����3��80%��
����3��80%��
��4��80����5��22.6%����6��32t
��������
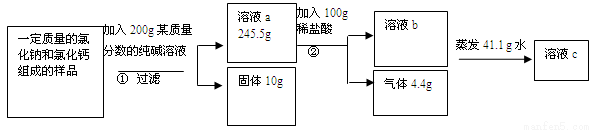
�����������μӷ�Ӧ��̼���Ƶ�����Ϊx �Ȼ���ΪY�����ɵ��Ȼ��Ƶ�����ΪZ
CaCl2+Na2CO3===2NaCl+CaCO3��
111 106 117 100
Y x z 10��
�μӷ�Ӧ�Ĵ������� (X)�ı���ʽΪ =
= ���x=10.6�ˣ������������Y=11.1�� z=11.7�ˣ�
���x=10.6�ˣ������������Y=11.1�� z=11.7�ˣ�
�����ɶ�����̼������Ϊ4.4����Ҫ����W��
Na2CO3+ 2HCl =2 NaCl+ CO2��+H2O
73 44
W 4.4�� �б���ʽ�ã�73��w=44��4.4�� ��ã�w=7.3��
����ϡ���������ʵ���������Ϊ=7.3��100��100%=7.3%��
���������غ㶨�ɿ���֪��ԭ����������=245.5��+10��-200��=55.5�� ������ԭ��������Ȼ��Ƶ���������Ϊ=44.4��55.5��100%=80%�����յ���Һ������Ϊ�Ȼ��ƣ���������=11.7��+44.4��=56.1�ˣ���c��Һ�����ʵ���������Ϊ=56.1�ˣ���245.5+100-4.4-41.4����100%=22.6%����Ҫ��36.5%��Ũ���������ΪE,�� E��36.5%=100��7.3% E=20�ˡ��������ˮ������Ϊ=100-20=80�ˣ��������������Ƶ�����Ϊr,
2NaCl + 2H2O 2NaOH + Cl2 ��+ H2��
2NaOH + Cl2 ��+ H2��
117 80
58.5t ��80% r
�б���ʽ�ã�117����58.5t��80%��=80��r ���r=32t
���㣺�����غ㶨�� ���ʵ����������ļ��� ��Һ��ϡ�� ���ݻ�ѧ����ʽ�ļ���

 ��У����ϵ�д�
��У����ϵ�д���ȥ���и������е��������ʣ����÷��������е���
ѡ�� | ���� | ���� | ��ȥ���ʵķ��� |
A | Cu�� | Fe�� | ������ |
B | NaCl | ��ɳ | ��ˮ�ܽ⡢���ˡ����� |
C | NaOH | Na2CO3 | ��������ϡ���������ٲ������� |
D | O2 | ˮ���� | ͨ��ʢ��Ũ�����ϴ��ƿ |
��������ʵ���۽��ʹ������
ѡ�� | �� ʵ | �� �� |
A | һ����̼�ж���������̼�� | �������ʵķ��ӹ��ɲ�ͬ |
B | Ʒ������ˮ����ɢ�ٶȸ��� | ��������£������˶����ʼӿ� |
C | ���ᡢϡ���ᶼ��ʹ��ɫʯ����Һ��� | ������Һ�ж����������� |
D | �¶ȼ��еľƾ��������� | ԭ�Ӽ�ļ������ʱ���� |
 �������йظƵ��жϴ������
�������йظƵ��жϴ������
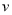 ��CH4����
��CH4���� ��O2������5��2
��O2������5��2 CO2 + 2H2O
CO2 + 2H2O