��Ŀ����
��������Һ����������������ͬ������Ħ������ΪM g/mol�������±���Ϣ���ش��й����⣺
| ���ʵ��������� | ��Һ���ܶ� | |
| ��һ����Һ | ��1 | ��1 |
| �ڶ�����Һ | ��2 | ��2 |
��2��ȡ������������Һ��ϣ������û��Һ�����ʵ�����������3=______��
��3�����Ц�1����2��ȡ�������������Һ��ϣ������û��Һ�����ʵ���������Ϊ��4���ԱȽϦ�3����4����Դ�С��______��
���𰸡���������1���������ʵ���Ũ��= ���м��㣮
���м��㣮
��2�������������������ļ��㹫ʽ���ֱ��������ʵ������ܼ��������м��㼴�ɣ�
��3��������һ�������⣬����ʱ�������4�ı���ʽ���3�Ƚϣ������ѧ�Ϲ��ڱȽϴ�С�ķ���������ɣ�
����⣺������Һ�����Ϊ1L�����ʵ�����Ϊm1�������ʵ���Ϊn1����Ũ��ΪC1������֪�����ʵ���������Ϊ��1����Һ���ܶȣ�g?cm-3��Ϊ��1����
�� 1����һ����Һ�У����ʵ����ʵ���Ũ��Ϊ��
m1=��1×V×W1×1000��n1=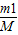
�� C1=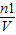 =
=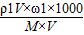 =
=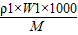
�ʴ�Ϊ��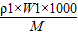
2���� ������Һ�����������ʷֱ�ΪM1��M2����
M1=m��1��M2=m��2
��3=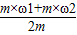 ×100%=��
×100%=��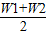 ��×100%��
��×100%��
ȡ������������Һ��ϣ������û��Һ�����ʵ�����������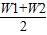 ��×100%
��×100%
�ʴ�Ϊ����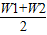 ��×100%
��×100%
��3��ȡ�������������Һ��ϣ����������ΪV��
����Һ�������ֱ�ΪM3 M4����
��ô�����ʵ������ֱ�Ϊm3 m4��
��4=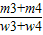 ×100%=
×100%=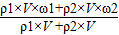
��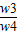 =
= +
+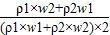 ����1����2�� ������1����2�����4����3��
����1����2�� ������1����2�����4����3��
����1����2�����3����4��
�ʴ�Ϊ������1����2�����4����3��
����1����2�����3����4��
������������һ�������������������Ŀ����⣬����Ĺؼ������úü�������ܶȼ���������������ת�����Ѷ����ڵ����ʵ���ѧ�Ƶ����Ƚϴ�С�����������ʵ���Ŀ��
 ���м��㣮
���м��㣮��2�������������������ļ��㹫ʽ���ֱ��������ʵ������ܼ��������м��㼴�ɣ�
��3��������һ�������⣬����ʱ�������4�ı���ʽ���3�Ƚϣ������ѧ�Ϲ��ڱȽϴ�С�ķ���������ɣ�
����⣺������Һ�����Ϊ1L�����ʵ�����Ϊm1�������ʵ���Ϊn1����Ũ��ΪC1������֪�����ʵ���������Ϊ��1����Һ���ܶȣ�g?cm-3��Ϊ��1����
�� 1����һ����Һ�У����ʵ����ʵ���Ũ��Ϊ��
m1=��1×V×W1×1000��n1=
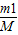
�� C1=
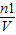 =
=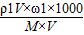 =
=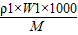
�ʴ�Ϊ��
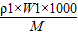
2���� ������Һ�����������ʷֱ�ΪM1��M2����
M1=m��1��M2=m��2
��3=
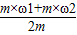 ×100%=��
×100%=��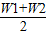 ��×100%��
��×100%��ȡ������������Һ��ϣ������û��Һ�����ʵ�����������
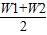 ��×100%
��×100%�ʴ�Ϊ����
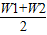 ��×100%
��×100%��3��ȡ�������������Һ��ϣ����������ΪV��
����Һ�������ֱ�ΪM3 M4����
��ô�����ʵ������ֱ�Ϊm3 m4��
��4=
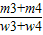 ×100%=
×100%=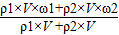
��
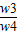 =
= +
+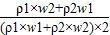 ����1����2�� ������1����2�����4����3��
����1����2�� ������1����2�����4����3������1����2�����3����4��
�ʴ�Ϊ������1����2�����4����3��
����1����2�����3����4��
������������һ�������������������Ŀ����⣬����Ĺؼ������úü�������ܶȼ���������������ת�����Ѷ����ڵ����ʵ���ѧ�Ƶ����Ƚϴ�С�����������ʵ���Ŀ��

��ϰ��ϵ�д�
�����Ŀ
ͬѧ��ѧϰ�ˡ���������һ�ºԽ������й����ʲ�����Ũ�����Ȥ��
̽��һ��ijС����������ͼ��ʾ��̽��ʵ�飮ʵ�鷢��CO��CuO���Ⱥ��ɫ��ĩ��ɺ�ɫ��ĩ����������С����о����̲�����������

[�о�����]̽����ɫ��ĩ����Ҫ�ɷ�
[��������]
��1���й����ʵ���ɫ��CuO��ĩ����ɫ��Cu2O��ĩ����ɫ
��2��CuO��Cu2O���ܺ�ϡ���ᷢ����Ӧ����ѧ����ʽΪ��
CuOʮH2SO4=CuSO4+H2O
Cu2O+H2SO4=CuSO4+Cu+H2O
[������ʵ��]
��1�����Ӳ�ʲ������ں�ɫ��ĩΪһ�����ʣ���������ijɷ֣�����Ƽ�ʵ��֤����IJ²⣮
��2�����Ӳ�ʲ������ں�ɫ��ĩΪ�������ʵĻ���Ҫȷ����ĩ���������ʵ���������������Ҫͨ���й����ݼ�������жϣ���������Ϊ�������ݵ��ǣ� ��
A����ӦǰCuO��ĩ��������B��Ӳ�ʲ������й������ʼ��ٵ�����
C��ͨ��CO��������D����Ӧ����������������
̽������ʵ����ͨ�����÷�Ӧ��Zn+H2SO4=ZnSO4+H2����������������
��1��̽��С�鷢��ʵ�������������ô�п�������ʵ�п�������ô�п��ԭ���Ǵ�п�����ᷴӦ�ų����������ʱȴ�п�죮�ֽ������������ݴ�п�۷ֱ����a��b�����ձ��У����ձ��о���ʢ����ͬ�Ĺ���ϡ���ᣬͬʱ��a�ձ��м���������CuO��ĩ����ַ�Ӧ�����ձ��У�
�ٲ���������������a b�������������=����
�ڲ������������ʣ�a b�������������=����
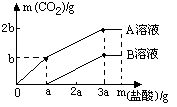
��2��Ϊ�˲ⶨ��ͭ��ͭ��п�Ͻ𣩵���ɣ�ij�о���ѧϰС���ȡ����Ʒ10g����������μ���9.8%��ϡ�������պò��ٲ�������Ϊֹ����Ӧ���������ɵ�����������������Һ��������ϵ����ͼ��ʾ���Լ��㣺
����Ʒ��ͭ����������Ϊ�� ��
��ǡ����ȫ��Ӧʱ������ϡ���������Ϊ �ˣ�
̽��һ��ijС����������ͼ��ʾ��̽��ʵ�飮ʵ�鷢��CO��CuO���Ⱥ��ɫ��ĩ��ɺ�ɫ��ĩ����������С����о����̲�����������

[�о�����]̽����ɫ��ĩ����Ҫ�ɷ�
[��������]
��1���й����ʵ���ɫ��CuO��ĩ����ɫ��Cu2O��ĩ����ɫ
��2��CuO��Cu2O���ܺ�ϡ���ᷢ����Ӧ����ѧ����ʽΪ��
CuOʮH2SO4=CuSO4+H2O
Cu2O+H2SO4=CuSO4+Cu+H2O
[������ʵ��]
��1�����Ӳ�ʲ������ں�ɫ��ĩΪһ�����ʣ���������ijɷ֣�����Ƽ�ʵ��֤����IJ²⣮
| ���� | ��ʵ�鷽�� | ���� | CO��CuO��Ӧ�Ļ�ѧ����ʽ |
| ��ĩΪCu | ����Ӧ | CuO+CO=Cu+CO2 | |
| ��ĩ�ܽ⣬��Һ�����ɫ�����ɺ�ɫ��ĩ | 2CuO+CO=Cu2O+CO2 |
A����ӦǰCuO��ĩ��������B��Ӳ�ʲ������й������ʼ��ٵ�����
C��ͨ��CO��������D����Ӧ����������������
̽������ʵ����ͨ�����÷�Ӧ��Zn+H2SO4=ZnSO4+H2����������������
��1��̽��С�鷢��ʵ�������������ô�п�������ʵ�п�������ô�п��ԭ���Ǵ�п�����ᷴӦ�ų����������ʱȴ�п�죮�ֽ������������ݴ�п�۷ֱ����a��b�����ձ��У����ձ��о���ʢ����ͬ�Ĺ���ϡ���ᣬͬʱ��a�ձ��м���������CuO��ĩ����ַ�Ӧ�����ձ��У�
�ٲ���������������a
�ڲ������������ʣ�a
��2��Ϊ�˲ⶨ��ͭ��ͭ��п�Ͻ𣩵���ɣ�ij�о���ѧϰС���ȡ����Ʒ10g����������μ���9.8%��ϡ�������պò��ٲ�������Ϊֹ����Ӧ���������ɵ�����������������Һ��������ϵ����ͼ��ʾ���Լ��㣺

����Ʒ��ͭ����������Ϊ��
��ǡ����ȫ��Ӧʱ������ϡ���������Ϊ
��������Һ����������������ͬ������Ħ������ΪM g/mol��
�����±���Ϣ���ش��й����⣺
��1����һ����Һ�У����ʵ����ʵ���Ũ��Ϊ ��
��2��ȡ������������Һ��ϣ������û��Һ�����ʵ�����������3= ��
��3�����Ц�1����2��ȡ�������������Һ��ϣ������û��Һ�����ʵ���������Ϊ��4���ԱȽϦ�3����4����Դ�С�� ��
�����±���Ϣ���ش��й����⣺
| ���ʵ��������� | ��Һ���ܶȣ�g?cm-3�� | |
| ��һ����Һ | ��1 | ��1 |
| �ڶ�����Һ | ��2 | ��2 |
��2��ȡ������������Һ��ϣ������û��Һ�����ʵ�����������3=
��3�����Ц�1����2��ȡ�������������Һ��ϣ������û��Һ�����ʵ���������Ϊ��4���ԱȽϦ�3����4����Դ�С��
 ��2012?��Ӫ����Ӫ�мƻ���5��ʱ�䣬���ֽ���30����̬�ֳ���ʵ�ֳ���ɭ�ֻ������غ���ˮ������Ŀ�꣮Ϊ��֤��ֲ��ľ�ijɻ��ʣ����ϻ����˵�ƿ������ͼ����ij����С����Ե�ƿ����Һ��ɷֽ���̽�����쿴��ƿ����˵���ܼ�û�о���ijɷ֣��������жϿ϶���һ��ֲ��Ӫ��Һ����������������������ϣ�
��2012?��Ӫ����Ӫ�мƻ���5��ʱ�䣬���ֽ���30����̬�ֳ���ʵ�ֳ���ɭ�ֻ������غ���ˮ������Ŀ�꣮Ϊ��֤��ֲ��ľ�ijɻ��ʣ����ϻ����˵�ƿ������ͼ����ij����С����Ե�ƿ����Һ��ɷֽ���̽�����쿴��ƿ����˵���ܼ�û�о���ijɷ֣��������жϿ϶���һ��ֲ��Ӫ��Һ����������������������ϣ�