��Ŀ����
7��2010��4��14���ຣ��������7.1��������������ˮ��ȫ���ܹ�ע����1����ȥˮ�������Թ������ʵIJ����ǹ��ˣ�
��2�������м��ܽ���ˮ��Ӳ�ȣ�����ɱ�������ķ����Ǽ�����У�
��3����84����Һ���������������ߣ��Ʊ�����Ҫ�ɷִ������ƣ�NaClO���Ļ�ѧ����ʽΪ2NaOH+X�TNaClO+NaCl+H2O����X�Ļ�ѧʽ��Cl2��
��4����ˮ������֮Դ�������й���ˮ���������������õ�˵������ȷ����B��
A��ˮ���л���ܸ�������
B��ˮ��һ�ֺܺõ��ܼ�����ά���������Һƽ��
C��ˮ��������Ҫ�Ĺ�������
D��ˮ����Ԫ�غ�����ߣ����ṩ���������������
���� ��1�����ݹ����Ƿ��벻���Թ����Һ��ķ������з�����
��2�����ݶԾ���ˮ���÷�������ʶ���жϼ��ܽ���ˮ��Ӳ������ɱ�������ľ���ˮ�IJ���������
��3�����ݻ�ѧ��Ӧǰ��Ԫ�ص������ԭ�ӵĸ���������з�����
��4������ˮ����ɡ����ʼ����˵����ã��ж϶���ˮ��˵������ȷ��˵����
��� �⣺��1����ȥˮ�������Թ������ʣ�����ʹ�ù��˵ķ�����������ˣ�
��2��Ӳˮ�к��н϶�Ŀ����Ը�þ�����������ת���ɲ����Ը�þ������Ӷ�����ˮ��Ӳ�ȣ�������л���ɱ��ˮ�е�ϸ�������������У�
��3��2NaOH+X�TNaClO+NaCl+H2O����Ӧ����2��Naԭ�ӣ�2��Clԭ�ӣ�2��Hԭ�ӣ�2��Oԭ�ӣ���Ӧǰ��2��Naԭ�ӣ�2��Hԭ�ӣ�2��Oԭ�ӣ���X�к�������Clԭ�ӣ��仯ѧʽΪCl2�����Cl2��
��4��A��ˮ��H��O����Ԫ����ɵĻ��������в���CԪ�أ��������л����A����ȷ��
B���������ʿ����ܽ���ˮ�У�ˮ��һ�ֺܺõ��ܼ�����ά���������Һƽ�⣻��B��ȷ��
C������Ӫ�����������ࡢ�����ʡ�֬�������л�����ǹ���ϸ���ijɷ֣�����Ϊ������ṩ��������ˮ�����ṩ����������������C����ȷ��
D��ˮ����Ԫ�غ����ܸ߿ɴ��90%����ˮ�в�����������Ҳ�����������зֽ⣬��˲����ṩ�����������������D����ȷ��
���B��
���� ����Ϊ����ˮ�й�֪ʶ�Ŀ��飬��Ŀ�ѶȲ����漰֪ʶ��϶࣬�������ڹ��ɡ��Աȵ�ϰ�߶��ڽ������������нϴ������

 ���Ž�������С״Ԫϵ�д�
���Ž�������С״Ԫϵ�д�| A�� | 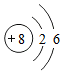 | B�� | 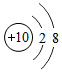 | C�� | 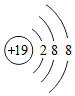 | D�� | 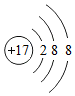 |
CuO+H2SO4�TCuSO4+H2O��ͭ���μӷ�Ӧ��
| ��һ�� | �ڶ��� | ������ | ���Ĵ� | ����� | |
| ����ϡ�����������g�� | 20 | 20 | 20 | 20 | 20 |
| ��ַ�Ӧ��ʣ������������g�� | 8 | 6 | 4 | 2 | 2 |
��1��10g��Ʒ�У�����ͭ������Ϊ8g��
��2��10g��Ʒ��ͭԪ�ص������ܹ�Ϊ���ٿˣ�
��3������ϡ������10g����Ʒ��ȫ��Ӧ�����ɶ��ٿ�����ͭ��