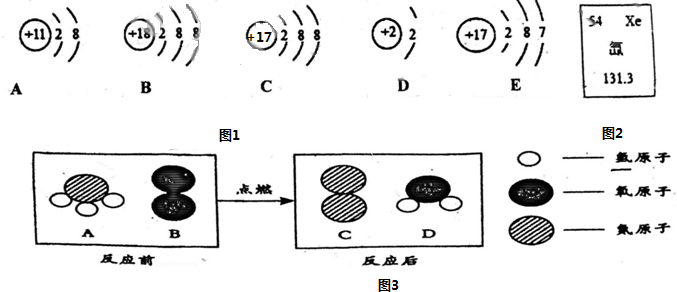
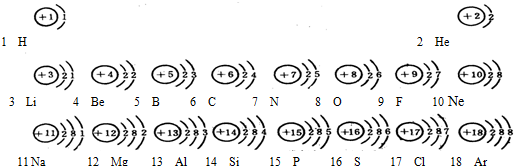
��Ŀ����
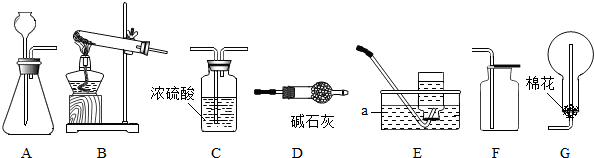
4����ͼ��ʵ�����Ʊ�����ij���װ�ã���ش��������⣮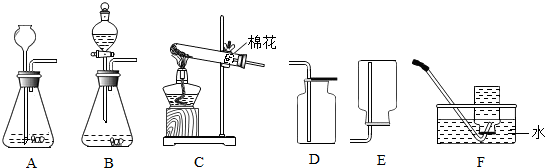
��1��Aװ���г�Ϸ��©����������Һ�����µ�ԭ���Ƿ�ֹ����ӳ���©���ݳ���
��2���������װ����ѡ��ʵ�����ù���������Һ�Ͷ���������ȡ������һ�鷢�����ռ�װ�ã�AD������ĸ�����÷�Ӧ�Ļ�ѧ����ʽ��2H2O2$\frac{\underline{\;MnO_2\;}}{\;}$2H2O+O2�������������̼������ռ����ķ����ǽ�ȼ�ŵ�ľ�����ڼ���ƿ�пڣ���ľ��Ϩ��֤�����ռ�����
��3����ʵ�����ù�������Ͷ���������ȡ������ѡ��ķ���װ�ú��ռ�װ��ΪAD��AF��BD��BF������ĸ������������̵������Ǵ����ã�
���� ��1��������ȡ�����Ƕ�װ�õ�Ҫ������ش�
��2��������ȡ������ҩƷ״̬�ͷ�Ӧ����ѡ��װ�ã����ݷ�Ӧ������P�����غ㶨�ɷ��������ݶ�����̼��֧��ȼ�յ�������������
��3��������ȡ������ҩƷ״̬�ͷ�Ӧ����������������ѡ��װ�ã����ݶ������̵Ĵ����÷�����
��� �⣺��1������ȡ����ʱ������©���¶˱�������Һ�����£�ԭ���Ƿ�ֹ�����ܵ���
��2������������Һ�Ͷ��������ڳ����¾��ܷ�Ӧ��������������ˮ���ܶȱȿ�����������ˮ���������ſ����ռ�����ѡ�õ�װ��Ϊ��AD��AF��BD��BF����ѧ����ʽΪ2H2O2$\frac{\underline{\;MnO_2\;}}{\;}$2H2O+O2�����ռ�������̼ʱ�����������ռ����ķ����ǽ�ȼ�ŵ�ľ�����ڼ���ƿ�пڣ���ľ��Ϩ��֤�����ռ�����
��3����ʵ�����ù�������Ͷ���������ȡ������ѡ��ķ���װ�ú��ռ�װ��ΪAD��AF��BD��BF���������̵������Ǵ����ã�
�ʴ�Ϊ��
��1����ֹ����ӳ���©���ݳ���
��2��AD��2H2O2$\frac{\underline{\;MnO_2\;}}{\;}$2H2O+O2������ȼ�ŵ�ľ�����ڼ���ƿ�пڣ���ľ��Ϩ��֤�����ռ�����
��3��AD��AF��BD��BF�������ã�
���� ���⿼����ʵ������ȡ�����Ͷ�����̼�ķ�Ӧԭ����װ�ã�������֪ʶ������֪ʶ���Ӧ�õ������У�

 �������Ӧ���⼯ѵϵ�д�
�������Ӧ���⼯ѵϵ�д�| A�� | C+O2$\frac{\underline{\;��ȼ\;}}{\;}$CO2 | B�� | 2H2O$\frac{\underline{\;ͨ��\;}}{\;}$2H2��+O2�� | ||
| C�� | 3CO+Fe2O3$\frac{\underline{\;����\;}}{\;}$2Fe+3CO2 | D�� | BaCl2+Na2SO4�TBaSO4��+2NaCl |
| A�� | 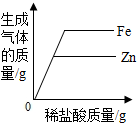 �ֱ����������п�����еμ������������������ | |
| B�� | 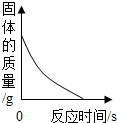 ���������£���һ������������ͨ��һ����̼���� | |
| C�� | 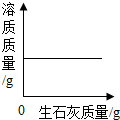 һ���¶��£���һ�����ı���ʯ��ˮ�в��ϼ�����ʯ�� | |
| D�� | 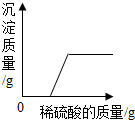 ��һ�������������ƺ����ᱵ�Ļ����Һ�еμ�ϡ���� |
 �����ѡ������ͼ��ʾ����������ǣ�������
�����ѡ������ͼ��ʾ����������ǣ�������| ��� | ������ | ������ |
| A | ��һ����ϡ�����м���п�۵����� | ��Һ����Ԫ�ص����� |
| B | ��һ�������������ƺ�̼���ƻ����Һ�м���ϡ��������� | ������������� |
| C | ��һ�����ij���ʯ��ˮ�м���̼������Һ������ | ���ɳ��������� |
| D | ��һ���¶��£���һ�����������������Һ�м�������ع�������� | ��Һ�����ʵ��������� |
| A�� | A | B�� | B | C�� | C | D�� | D |
| A�� | ��ȥNaOH��Һ�е�Ca��OH��2���ʣ���������Na2CO3��Һ������ | |
| B�� | ��ε��ʲ�����������ʻ��ã���������������ɷ�Ч��ʧ | |
| C�� | NaOH+HCl�TNaCl+H2O�����ڸ��ֽⷴӦ | |
| D�� | ���ʯ��ʯī�������ʲ�ͬ��ԭ���ǹ������ǵ�ԭ�ӽṹ��ͬ |
 Ϊ���ٻ�ʯȼ�ϵ�ʹ�ã�������Ⱦ���������ڻ������м������£�
Ϊ���ٻ�ʯȼ�ϵ�ʹ�ã�������Ⱦ���������ڻ������м������£�