��Ŀ����
ͭ�Ͻ��кܶ����࣮��������ͭп�Ͻ�ͭ���Ͻ�ͭ���Ͻ�ͭ���Ͻ�����һ��ͭ�Ͻ�Ϊȷ����Ϊ���ֺϽ�����һ�֣�������ʵ�飺��20�˺Ͻ���Ʒ�гɷ�ĩ���ֳɵ����������ݣ�����һ�ݼ��뵽������20��ϡ�����У��Ͻ��ܽ⣬�����ݲ�������Ӧ����ˣ��ù���3.5�ˣ���һ�ݼ��뵽49.9������ͭ��Һ�У���ַ�Ӧ����ˣ��õ���ɫ��Һ����9.9�ˣ���������˵�ϡH2SO4�в��ܽ⣮
��1���úϽ�ijɷ�Ϊ______��
��2�����뵽����ͭ��Һ�з�����Ӧ�Ļ�ѧ����ʽΪ______��
��3����������ͭ��Ӧ�Ľ���������x���ı���ʽΪ______��
��4���Ͻ���Ʒ��ͭ����������Ϊ______��
��5��������ͭ��Һ��Ӧ��������ɫ��Һ��������������Ϊ______��
��1���úϽ�ijɷ�Ϊ______��
��2�����뵽����ͭ��Һ�з�����Ӧ�Ļ�ѧ����ʽΪ______��
��3����������ͭ��Ӧ�Ľ���������x���ı���ʽΪ______��
��4���Ͻ���Ʒ��ͭ����������Ϊ______��
��5��������ͭ��Һ��Ӧ��������ɫ��Һ��������������Ϊ______��
��1����������Һ�ʻ�ɫ����������Һ�ʺ�ɫ������п��Һ����ɫ���ʴ�Ϊ��ͭп�Ͻ�
��2��п������ͭ��Һ������Ӧ�Ļ�ѧ����ʽΪ��Zn+CuSO4=ZnSO4+Cu���ʴ�Ϊ��Zn+CuSO4=ZnSO4+Cu��
��3����������ͭ��Ӧ�Ľ���п������Ϊx��
��������ͭ��Ӧ�Ľ���������x���ı���ʽΪ��65��64=x����9.9g-3.5g����
�ʴ�Ϊ��65��64=x����9.9g-3.5g����
��4���Ͻ���Ʒ��ͭ����������Ϊ��
��100%=35%���ʴ�Ϊ��35%��
��5����������ͭ��Һ��Ӧ��������ɫ��Һ����������Ϊy��
Zn+CuSO4=ZnSO4+Cu
65 161
9.9g-3.5g y
��65��161=��9.9g-3.5g ����y��
��֮�ã�y=15.85g��
������ͭ��Һ��Ӧ��������ɫ��Һ����Ϊ��49.9g+10g-9.9g=50g��
������ͭ��Һ��Ӧ��������ɫ��Һ��������������Ϊ��
��100%=31.7%��
�ʴ�Ϊ��31.7%��
��2��п������ͭ��Һ������Ӧ�Ļ�ѧ����ʽΪ��Zn+CuSO4=ZnSO4+Cu���ʴ�Ϊ��Zn+CuSO4=ZnSO4+Cu��
��3����������ͭ��Ӧ�Ľ���п������Ϊx��
��������ͭ��Ӧ�Ľ���������x���ı���ʽΪ��65��64=x����9.9g-3.5g����
�ʴ�Ϊ��65��64=x����9.9g-3.5g����
��4���Ͻ���Ʒ��ͭ����������Ϊ��
| 3.5g��2 |
| 20g |
��5����������ͭ��Һ��Ӧ��������ɫ��Һ����������Ϊy��
Zn+CuSO4=ZnSO4+Cu
65 161
9.9g-3.5g y
��65��161=��9.9g-3.5g ����y��
��֮�ã�y=15.85g��
������ͭ��Һ��Ӧ��������ɫ��Һ����Ϊ��49.9g+10g-9.9g=50g��
������ͭ��Һ��Ӧ��������ɫ��Һ��������������Ϊ��
| 15.85 |
| 50 |
�ʴ�Ϊ��31.7%��

��ϰ��ϵ�д�
 �����Ļ���������人������ϵ�д�
�����Ļ���������人������ϵ�д� ���������ּ���ÿһ��ȫ�º�����ҵ��ϵ�д�
���������ּ���ÿһ��ȫ�º�����ҵ��ϵ�д� ��ٽ������½������������ϵ�д�
��ٽ������½������������ϵ�д�
�����Ŀ
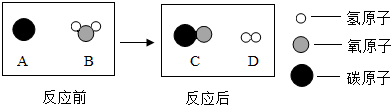


 �ˡ����������������ǵ� ��������ʹ�������������������еĹ�ϵ�����������Լ����������ֽ�����̽�����ǵĽ������˳���ܴﵽĿ���� ������ţ���
�ˡ����������������ǵ� ��������ʹ�������������������еĹ�ϵ�����������Լ����������ֽ�����̽�����ǵĽ������˳���ܴﵽĿ���� ������ţ���