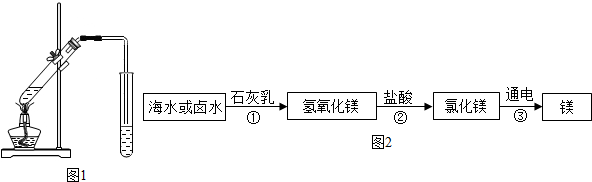
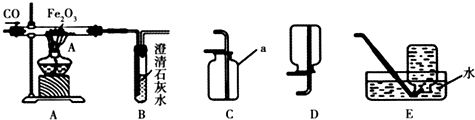��Ŀ����
14��NaOH��Na2CO3��NaCl�ڲ�ͬ�ܼ��е��ܽ�������ʾ����1����NaOH��Na2CO3��NaCl�ֱ���ˮ�е��ܽ�ȣ�S/g��
| t/�� | NaOH | Na2CO3 | NaCl |
| 0 | 42 | 7.1 | 35.7 |
| 10 | 51 | 12.2 | 35.8 |
| 20 | 109 | 21.8 | 36.0 |
| 30 | 119 | 39.7 | 36.3 |
| NaOH | Na2CO3 | NaCl |
| 17.3 | ��0.01 | 0.1 |
��1��NaOH��ˮ�е��ܽ�����¶����߶����������С����
��2��20��ʱ������ʳ��ˮ�����ʵ���������Ϊ26.5%����������ȷ��0.1%����
��3��Ϊ֤��CO2����NaOH������Ӧ��С��ͬѧ��CO2ͨ�뱥��NaOH���Ҵ���Һ�У����Ʋ��ʵ��ɹ۲쵽�������а�ɫ�������ɣ��Ʋ�������̼�������Ҵ��е��ܽ�Ⱥ�С��
���� ��1������NaOH��ˮ�е��ܽ�����ݣ����з������
��2�����ݱ�����Һ��������������=$\frac{�ܽ��}{�ܽ��+100g}$��100%�����з������
��3������CO2��NaOH��Ӧ����̼���ƺ�ˮ��̼�������Ҵ������ܣ����з������
��� �⣺��1����NaOH��ˮ�е��ܽ�����ݣ�NaOH��ˮ�е��ܽ�����¶����߶�����
��2��20��ʱ���Ȼ��Ƶ��ܽ��Ϊ36.0g������ʳ��ˮ�����ʵ���������Ϊ$\frac{36.0g}{36.0g+100g}$��100%��26.5%��
��3��CO2��NaOH��Ӧ����̼���ƺ�ˮ��̼�������Ҵ��е��ܽ�Ⱥ�С���ʿɹ۲쵽���������а�ɫ�������ɣ�
�ʴ�Ϊ����1������2��26.5%����3���а�ɫ�������ɣ�̼�������Ҵ��е��ܽ�Ⱥ�С��
���� �����ѶȲ������ճ������ʵ��ܽ�����¶�Ӱ��ı仯���ơ�������Һ��������������=$\frac{�ܽ��}{�ܽ��+100g}$��100%������ȷ�����Ĺؼ���

��ϰ��ϵ�д�
�����Ŀ
2�������������ʵķ�������ȷ���ǣ�������
| A�� | �÷�̪��Һ����ϡ�����ʳ��ˮ | |
| B�� | ��ȼ�ŵ�ľ�����������Ͷ�����̼ | |
| C�� | �÷���ˮ����Ӳˮ����ˮ | |
| D�� | ��ˮ��������粒�����������ƹ��� |
3����ѧ��Ӧ����һЩ��ȫ��ʶ�������й�������˵����ȷ���ǣ�������
| A�� | ù��Ĵ����������Է���ʳ�� | |
| B�� | Ϊ���õ簲ȫ�����Ӵ���ѹ�����壬��������ѹ������ | |
| C�� | ȼ���ŵľƾ��Ʊ�������Ҫ���촵���Է�ֹ�����ľƾ�ȼ������ | |
| D�� | Ϊ��ֹ�����·���õ������ܹ��ʹ����նϱ���˿������ͣ���¹ʣ�Ҫ���ýϴֵı���˿ |

 ���������ǵ���������˷��㣬��ͼΪ��ͨ������ʾ��ͼ��
���������ǵ���������˷��㣬��ͼΪ��ͨ������ʾ��ͼ��