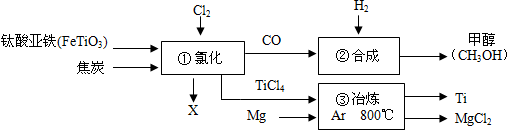
��Ŀ����
3����1�������ֺͷ�����գ���������������2N2��������������OH-���۱������þ��þԪ�صĻ��ϼ�$\stackrel{+2}{Mg}$O��
��2����˵�����з��ŵĺ��壺
��2N2����ԭ�ӣ���3O23�������ӣ���4Al3+4�������ӣ�
��3��DZˮͧ�п��ù������ƣ�Na2O2����Ϊ������Դ���仯ѧ��Ӧ����ʽΪ��
2Na2O2+2CO2=2Na2CO3+X�������������غ㶨���ƶϣ�X�Ļ�ѧʽ��O2��
��4��д�����з�Ӧ�Ļ�ѧ����ʽ��
�����ڿ�����ȼ��4P+5O2$\frac{\underline{\;��ȼ\;}}{\;}$2P2O5���÷�Ӧ�Ļ�ѧ��Ӧ����Ϊ���Ϸ�Ӧ��
�����������Fe2O3+6HCl�T2FeCl3+3H2O��
���� ��1��ÿ����������2����ԭ�ӹ��ɣ�
ÿ�����������Ӵ�1����λ����ɣ�
Ԫ�صĻ��ϼ�Ӧ�ñ���Ԫ�ط��ŵ����Ϸ���
��2����ͬ�ķ��ű�ʾ��ͬ�����壻
��3����ѧ��Ӧǰ��Ԫ�ص�����䣬ԭ�ӵ����ࡢ�ܸ������䣻
��4�����ڿ�����ȼ���������������ף�
�������Ҫ�ɷ������������ܺ�ϡ���ḺӰ�ӳ��Ȼ�����ˮ��
��� �⣺��1���������������ӿ��Ա�ʾΪ2N2��
�����������ӿ��Ա�ʾΪOH-��
������þ��þԪ�صĻ��ϼ���+2�����Ա�ʾΪ$\stackrel{+2}{Mg}$O��
���2N2��OH-��$\stackrel{+2}{Mg}$O��
��2����2N��ʾ2����ԭ�ӣ�
��3O2��ʾ3�������ӣ�
��4Al3+��ʾ4�������ӣ�
���2����ԭ�ӣ�3�������ӣ�4�������ӣ�
��3��2Na2O2+2CO2=2Na2CO3+X�������������غ㶨�ɿ����ƶϳ�X�������������Ļ�ѧʽ��O2��
���O2��
��4�������ڿ�����ȼ�յĻ�ѧ����ʽΪ��4P+5O2$\frac{\underline{\;��ȼ\;}}{\;}$2P2O5�����ڻ��Ϸ�Ӧ��
�����������Ļ�ѧ����ʽΪ��Fe2O3+6HCl�T2FeCl3+3H2O��
���4P+5O2$\frac{\underline{\;��ȼ\;}}{\;}$2P2O5�����Ϸ�Ӧ��Fe2O3+6HCl�T2FeCl3+3H2O��
���� ������Ҫ����ѧ���Ի�ѧ�������д��������������Ŀ�ص㿼����ѧ���Ի�ѧ���ŵ���д������ȫ�棬ע�ػ�������Ŀ�ѶȽ�С��

| A�� | 10mL�ƾ���10mLˮ��Ϻ����С��20mL������Ϊ���ӱ�С�� | |
| B�� | NH4NO3��������ʯ��ͬʱʩ�� | |
| C�� | �������ס��������еġ������͡��ơ�ָ����ԭ�� | |
| D�� | ��ѧ��ÿ��ȵ�ţ����һ����Һ |
| A�� | ������ˮ����������������������� | |
| B�� | �÷�̪��Һ�����Ȼ�����Һ��ϡ���� | |
| C�� | ��ʯ����Һ����ϡ�ռ���Һ��ʯ��ˮ | |
| D�� | �ü�ϡ����ķ���������˿����˿ |
| A�� | C2O3���ȼ�յIJ�����CO2 | B�� | C2O3��ʹ����ʯ��ˮ����� | ||
| C�� | ��һ��������C2O3�ܻ�ԭ������ | D�� | C2O3��̼�Ļ��ϼ�Ϊ+3�� |
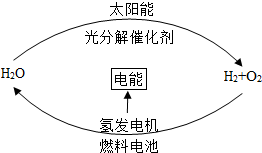 ��ͼ��һ��������Ļ������Դ��ѭ����ϵ��ȼ�ϵ���з�����Ӧ�Ļ�ѧ����ʽ��2H2+O2$\frac{\underline{\;��ȼ\;}}{\;}$2H2O��ȼ�ϵ���ǽ���ѧ��ת��Ϊ���ܵ�װ�ã����ŵ�������Ⱦ��
��ͼ��һ��������Ļ������Դ��ѭ����ϵ��ȼ�ϵ���з�����Ӧ�Ļ�ѧ����ʽ��2H2+O2$\frac{\underline{\;��ȼ\;}}{\;}$2H2O��ȼ�ϵ���ǽ���ѧ��ת��Ϊ���ܵ�װ�ã����ŵ�������Ⱦ��