��Ŀ����
2����Դ������������ȫ���ע���ȵ����⣮��1�����dz��õ�ȼ�ϴ�����Ի�ʯȼ�ϣ���ú��ʯ�ͺ���Ȼ���ȣ���Ȼ������Ҫ�ɷ�ȼ�յĻ�ѧ����ʽΪCH4+2O2$\frac{\underline{\;��ȼ\;}}{\;}$CO2+2H2O
��2����ʯȼ�ϴ���ʹ�û����SO2�ȿ�����Ⱦ���Щ�����ڿ����з�Ӧ���������������ˮ�����γ����꣮��������Ĵ�ʩ��ҵ�����辻���������������ŷţ���һ�����ɣ�
��3���ֽΣ����Ǵ�������ȼ��ȼ�ղ�����������ij��ѧС�����о�ȼ�յ�����ʱ��������ͼ��ʾ��ʵ�飮

�ٸĽ����װ�ã���ͼ������ͼһ��ȣ����ŵ��Ǽ�����Ⱦ����������
�ڽ�װ��ij����Ĵ��Թܿڳ��´�ֱ����ˮ�У����Թ���ס���ף���ͼ����ʾ��������۲쵽�ˡ�ˮ�����ݵ���ۡ�������Թ���װ���������O2���ѧʽ����
���� ��1��ú��ʯ�ͺ���Ȼ�����ڻ�ʯȼ�ϣ��Dz���������Դ������ȼ������ˮ�Ͷ�����̼��
��2�����������ǵ��������һ�����ʣ�
��3���ٶԱ�ͼһ��ͼ����ͼ���������ܱյ��Թ��н���ʵ�飬���Է�ֹ���ɵ�������������ɿ�����Ⱦ��
�ڰ����¶ȴﵽ���Ż���û�ܺ������Ӵ���ȼ����Ҫ����֧�֣��Թ���Ӧ�ṩȼ����Ҫ��������
��� �⣺��1��Ŀǰ��������������Դ��ú��ʯ�ͺ���Ȼ���Ȼ�ʯȼ�ϣ���Ȼ���е���Ҫ�ɷ��Ǽ��飬����ȼ�յĻ�ѧ����ʽΪ��CH4+2O2$\frac{\underline{\;��ȼ\;}}{\;}$CO2+2H2O��
��2��úȼ�ջ����������CO2��SO2��CO�����壬���лᵼ�������������SO2�����δ�ʩΪ��ҵ�����辻���������������ŷţ���ֹ��ɴ�����Ⱦ���˷�����Դ��
��3����ͼ���������ܱյ��Թ��У����Է�ֹȼ�պ����ɵ�������������Ⱦ��������ͼһ��ȼ�ղ���ֱ�����������
���Թ���ӦΪ��������������������ȼ������Ҫ��������
�ʴ�Ϊ����������������
�𰸣�
��1��ʯ�ͣ�CH4+2O2$\frac{\underline{\;��ȼ\;}}{\;}$CO2+2H2O��
��2��SO2����ҵ�����辻���������������ŷţ�
��3���ټ�����Ⱦ����������
��O2
���� ��ʯȼ�ϵ�ȼ���ܹ����������Ķ��������������Щ�������γ��������Ҫ���ʣ���˼��ٻ�ʯȼ�ϵ�ʹ�ã��ۺ����û�ʯȼ�������ڼ�������IJ�����

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�| A�� | ǧ����⣬����ѩƮ�����ԡ�����•ѩ���� | |
| B�� | ���ϵ���˿����������ɻ���ʼ�ɣ����ԡ����⡷�� | |
| C�� | ����ֱ����ǧ�ߣ�������������죨���ԣ�����®ɽ�ٲ����� | |
| D�� | ����Ϊ�������������ԡ���¥�Ρ��� |
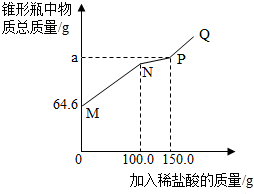 ȡ14.6g���ʵ��������ƹ�����Ʒ������ˮ������ƿ�У�����50.0gˮ������ܽ⣬������ƿ�еμӵ�������������Ϊ7.3%��ϡ���ᣮʵ���ü���ϡ�������������ƿ�����ʵ���������ϵ��ͼ��ʾ����̼������ϡ���ᷴӦ���������У�Na2CO3+HCl�TNaHCO3+NaCl��NaHCO3+HCl�TNaCl+H2O+CO2��������˵����ȷ���ǣ�������
ȡ14.6g���ʵ��������ƹ�����Ʒ������ˮ������ƿ�У�����50.0gˮ������ܽ⣬������ƿ�еμӵ�������������Ϊ7.3%��ϡ���ᣮʵ���ü���ϡ�������������ƿ�����ʵ���������ϵ��ͼ��ʾ����̼������ϡ���ᷴӦ���������У�Na2CO3+HCl�TNaHCO3+NaCl��NaHCO3+HCl�TNaCl+H2O+CO2��������˵����ȷ���ǣ�������| A�� | N�����Һ��ʾ�������Ʊ�ǡ����ȫ��Ӧ | |
| B�� | PQ�Σ�����P�㣩��Һ��pH��7 | |
| C�� | NP�α�ʾ��������Ĺ��̣�a��ֵΪ201.2 | |
| D�� | �ù�����Ʒ���������Ƶ�����Ϊ8.0g |
| A�� |  | B�� |  | C�� |  | D�� | 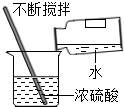 |
| ��һ�� | �ڶ��� | ������ | ���Ĵ� | |
| ��������������Һ�������ˣ� | 40 | 40 | 40 | 30 |
| ���ɳ��������������ˣ� | 0 | 4.9 | 9.8 | 9.8 |
��2��д�����ɳ����Ļ�ѧ����ʽCuSO4+2NaOH=Cu��OH��2��+Na2SO4
��3�����ݳ����������г���μӷ�Ӧ������ͭ������x���ı���ʽΪ$\frac{160}{x}$=$\frac{98}{9.8g}$
��4���û����Һ���������������Ϊ4.9%
��5������������ǡ����ȫ��Ӧʱ�����˺�������Һ����2.8gˮ��������������Ϊ10%
��6�������200�ַ�Һ����ȡ����ͭ���������Ͽ�����ȡ����ͭ������Ϊ12.8t��
��1�����ڣ��ҹ������Ի�ʯȼ��Ϊ��Ҫ��Դ��������������Ļ�ʯȼ�ϰ���ú��ʯ�ͺ���Ȼ���ȣ��������ڲ���������Դ����ɡ����ɡ�����
��2����ѧ��ӦҲ������������������з�Ӧ�����л�ų���������AC�������ţ�
A����ʯ����ˮ������������������B��̼�������̼������������������C��þ������
��3��Ϊ���ٻ�����Ⱦ�����ú�������ʣ��ɽ���ת��Ϊ��ȼ�����壬�˹��̿���Ϊ��̼��ˮ�ķ�Ӧ������ʾ��ͼ��ͼ��ʾ���÷�Ӧ�Ļ�ѧ����ʽΪC+H2O$\frac{\underline{\;һ������\;}}{\;}$CO+H2��������Ӧ����Ϊ�û���Ӧ��

��4��Ϊ�˼��ٻ�����Ⱦ���������壬����Ŭ�����ٶԻ�ʯȼ�ϵ����������о�����NH3ȼ�յIJ���Ҳû����Ⱦ�����ͷŴ�����������Ϊȼ������ijЩ���滹����H2��
�ٽ�NH3ȼ�շ�Ӧ�Ļ�ѧ����ʽ����������4NH3+3O2$\frac{\underline{\;��ȼ\;}}{\;}$6H2O+2N2��
��������±���Ϣ������NH3ȡ��H2���ŵ㣺
| ��ɫ����ζ | �۵㣨�棩 | �е㣨�棩 | ����ʱ��1Lˮ�����ܽ��������� | ������ܶȡ���g/L�� | |
| NH3 | ��ɫ���̼��� | -77.7 | -33 | 700L | 0.6942 |
| H2 | ��ɫ����ζ | -259.2 | -253 | 0.017L | 0.0899 |