��Ŀ����
���ݳ�����ѧ֪ʶ�������ͼ��ʾ�ش�
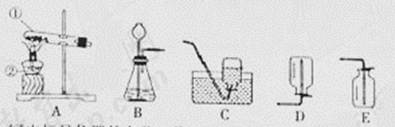
��1��д����Ţٵ��������ƣ�
��2��ʵ�����Ʊ����ռ�������̼����Ӧѡ�õ�װ����
��3��ʵ���ҳ��ü���NH4Cl��Ca��OH��2��������ķ�����ȡ������NH3��������ȡ�����ķ���װ��Ӧѡ��
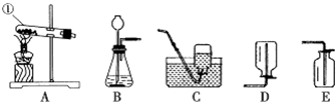
��1��д����Ţٵ��������ƣ�
�Թ�
�Թ�
����2��ʵ�����Ʊ����ռ�������̼����Ӧѡ�õ�װ����
BE
BE
����װ����ţ���������Ӧ�Ļ�ѧ����ʽ��CaCO3+2HCl�TCaCl2+H2O+CO2��
CaCO3+2HCl�TCaCl2+H2O+CO2��
����3��ʵ���ҳ��ü���NH4Cl��Ca��OH��2��������ķ�����ȡ������NH3��������ȡ�����ķ���װ��Ӧѡ��
A
A
����װ����ţ���
��������1���ݳ��������ش�
��2���ݷ�Ӧ��״̬�ͷ�Ӧ����ѡ����װ�ã��ݶ�����̼ �ܶȺ��ܽ���ѡ���ռ�װ�ã��ݷ�Ӧԭ����д����ʽ��
��3����ʵ������ȡ�����ķ�Ӧԭ���ͷ���װ�õ�ѡȡ�����������
��2���ݷ�Ӧ��״̬�ͷ�Ӧ����ѡ����װ�ã��ݶ�����̼ �ܶȺ��ܽ���ѡ���ռ�װ�ã��ݷ�Ӧԭ����д����ʽ��
��3����ʵ������ȡ�����ķ�Ӧԭ���ͷ���װ�õ�ѡȡ�����������
����⣺��1����Ţٵ��������Թܣ�
��2��ʵ�����Ʊ�������̼�ô���ʯ��ϡ���᳣�·�Ӧ��ȡ�����ڹ�Һ�����ͣ���ѡ����װ��B��������̼���ܶȱȿ�������������ˮ�����������ſ������ռ���������Ӧ�Ļ�ѧ����ʽ��CaCO3+2HCl�TCaCl2+H2O+CO2����
��3��ʵ���ҳ��ü���NH4Cl��Ca��OH��2��������ķ�����ȡ������NH3�������ڹ�������ͣ���ѡ����װ��A��
�ʴ�Ϊ����1���Թܣ� ��2��BE��CaCO3+2HCl�TCaCl2+H2O+CO2���� ��3��A��
��2��ʵ�����Ʊ�������̼�ô���ʯ��ϡ���᳣�·�Ӧ��ȡ�����ڹ�Һ�����ͣ���ѡ����װ��B��������̼���ܶȱȿ�������������ˮ�����������ſ������ռ���������Ӧ�Ļ�ѧ����ʽ��CaCO3+2HCl�TCaCl2+H2O+CO2����
��3��ʵ���ҳ��ü���NH4Cl��Ca��OH��2��������ķ�����ȡ������NH3�������ڹ�������ͣ���ѡ����װ��A��
�ʴ�Ϊ����1���Թܣ� ��2��BE��CaCO3+2HCl�TCaCl2+H2O+CO2���� ��3��A��
���������շ������ռ�װ�õ�ѡȡ������ʵ������ȡ������̼�ķ�Ӧԭ���������������ȣ�����˳�����

��ϰ��ϵ�д�
 ������״Ԫ��ҵϵ�д�
������״Ԫ��ҵϵ�д� ��ʱ�ƿ�������ϰϵ�д�
��ʱ�ƿ�������ϰϵ�д� һ��һ��һ��ͨϵ�д�
һ��һ��һ��ͨϵ�д� �㽭֮��ѧҵˮƽ����ϵ�д�
�㽭֮��ѧҵˮƽ����ϵ�д�
�����Ŀ