��Ŀ����
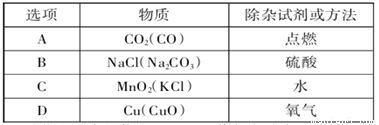
��һ�ܱ������У���X��O2��CO2��H2O�������ʣ���һ�������·���ij�ַ�Ӧ����Ӧһ��ʱ���÷�Ӧǰ������ʵ��������±�.����˵����ȷ����
���� | X | O2 | CO2 | H20 |
��Ӧǰ������/g | 19 | 34 | 6 | 2 |
��Ӧ�������/g | 11 | ���� | 28 | 20 |
A. ���еĴ���ֵΪ32 B. X��һ������C��H��OԪ��
C. �÷�Ӧ���û���Ӧ D. ��Ӧ��CO2��H2O��������Ϊ11:9
D ��������A�����������غ㶨�ɿ���֪��������ֵ=19+34+6+2-11-28-20=2������B���������ⷴӦ������22g�Ķ�����̼��18g��ˮ��������32g ��������8g��X��22g������̼�к�����Ԫ�ص�����Ϊ22g�� =16g�� 18gˮ�к���Ԫ�ص�����Ϊ18g�� =16g������������Ԫ�ص�����Ϊ16g+16g=32g����μӷ�Ӧ������������������ȣ�˵��X��һ��ֻ����̼��������Ԫ��...
��ϰ��ϵ�д�
�����Ŀ
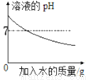
����ͼ������ȷ��ӳ��Ӧ�仯��ϵ����
|
|
|
|
A�������£�ϡ��KOH��Һ | B����Ũ����¶���ڿ����� | C����һ����ͭп�Ͻ��ĩ����μ���ϡ���� | D��һ�������ı���ʯ��ˮ�м�����ʯ�� |
A. A B. B C. C D. D
C ��������A����ˮϡ��KOH��Һ����Һ�����Ϊ���ԣ���pH����С��7������B����Ũ����¶���ڿ����л����տ����е�ˮʹ���ʵ�����������С��������Ϊ�㣬����C����һ����ͭп�Ͻ��ĩ����μ���ϡ���ᣬп����ϡ���ᷴӦ����ͭ���ܣ���ȷ��D����ʯ�Ҿ�����ˮ�ԣ����뵽����ʯ��ˮ�л�������Һ�е�ˮ��ʹ��Һ������С������ѡC��