��Ŀ����
��������(Na2O2)��һ�ֹ���������ˮ��Ӧ�Ļ�ѧ����ʽΪ2Na2O2��2H2O===4NaOH��O2������һ������Na2O2���뵽87.6gˮ�У���ȫ��Ӧ��������Һ�������ȷ�Ӧ���������������3.2g��ˮ�Ļӷ����Բ� �ƣ�������㣺
�ƣ�������㣺
��1����������������Ϊ_____g��
��2����Ӧ��������Һ�����ʵ�������
��3����������Һϡ�ͳ����ʵ���������Ϊ10%����Һ�����ˮ��������
��1��3.2
��2���⣺��μӷ�Ӧ�Ĺ������Ƶ�����Ϊx�������������Ƶ�����Ϊy��
2Na2O2��2H2O===4NaOH��O2��
��156������������160����32
��x���������� ����y���� 3.2g����
 ��
��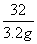 ��
��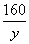 ��
��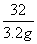
���x��15.6g��y��16g
��Ӧ��������Һ�����ʵ�����Ϊ16g
��3��16g/10% - (15.6+87.6-3.2) g=60 g
���ԡ�

 �����Ļ�������ҵϵ�д�
�����Ļ�������ҵϵ�д���ͬѧΪ���˽��������ͭ�Ľ������˳����������̽�����
�������ϣ�����Cr��������ɫ�й���Ľ������ڿ����б��������ɿ���ʴ�Ե���������Ĥ ��������ϡ���ᷴӦ���õ���ɫ�������Ǹ���CrSO4����Һ��
��������ϡ���ᷴӦ���õ���ɫ�������Ǹ���CrSO4����Һ��
ʵ����̣�ȡ��С��ͬ�����ֽ�����Ƭ����ɰֽ��ĥ������
| ʵ��һ | ʵ��� | ʵ���� | |
| ʵ�� ���� |
|
|
|
| ʵ�� ���� | ��Ƭ����������ݻ�������Һ�����ɫ�� |
| ��Ƭ����������ݽ� �졣 |
| ���� | ���ܺ�ϡ���ᷢ����Ӧ����Ӧ���ʽ����� | ͭ����ϡ���ᷴӦ |
|
| ���ֽ����Ļ��˳���� |
��1����ͬѧ���������ͭ��Һ��������ʵ���е�ϡ����Ҳ�ܵó����ֽ����Ļ��˳��ͬѧ��Ϊ�����У������� ��
��2����ͬѧ��ΪֻҪѡ������ҩƷ����ʵ��Ϳ��Եó����ֽ����Ļ��˳��������ҩƷ������ ��
Ϊ�˲ⶨij��Ʒ��̼�ᱵ��������������������ʵ�顣ȡһ��������Ʒ��400g������������Ϊ10%��ϡ��������ձ��С��ڻ�ѧ��Ӧ�����ж��ձ������е�ʣ�����������γ�������¼���±��������跴Ӧ���ٽ��У����ʲ�����ˮ��Ҳ����ϡ���ᷴӦ���ձ�������Ϊ25.4g��
| ��Ӧʱ�� | t1 | t2 | t3 | t4 | t5 |
| �ձ���ҩƷ����/g | 516.6 | 507.8 | m | 503.4 | 503.4 |
����㣺����Ӧ����ʽΪ��BaCO3 + 2HCl = BaCl2 + H2O + CO2����
��1�����еġ�m��Ϊ��
��2���ò�Ʒ��̼�ᱵ������������
��3������Ʒ��̼�ᱵ��ϡ����ǡ����ȫ��Ӧʱ��������Һ�����ʵ�����������
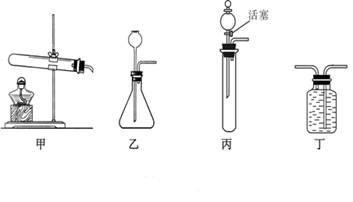
 ̼���ƺ�ϡ���ᷴӦ����ȡCO2�����ñ�װ��ȴ����˳���Ƶá�����װ����Ƚϣ���װ�õ������ŵ���_______________________���ô�װ�û�������ȡ������ ����ʽ�����ö�װ���ռ�����Ӧ�� �˽��롣��������ң�
̼���ƺ�ϡ���ᷴӦ����ȡCO2�����ñ�װ��ȴ����˳���Ƶá�����װ����Ƚϣ���װ�õ������ŵ���_______________________���ô�װ�û�������ȡ������ ����ʽ�����ö�װ���ռ�����Ӧ�� �˽��롣��������ң�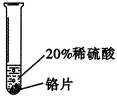
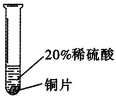
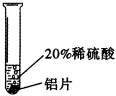
 ������Һ����������������
������Һ����������������