��Ŀ����
4����ͼ����������ȡ������ʵ��װ��ͼ��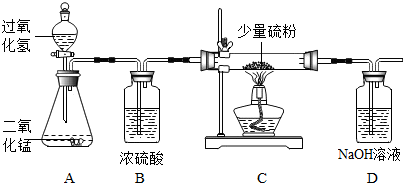
�ش��������⣺
��1��װ��B�������dz�ȥˮ�������������������ˮ�֣���װ��C�����ȼ�յ������Dz�������ɫ���棬װ��D�����������ն�������ֹ��Ⱦ������
��2��װ��A�з�����Ӧ�Ļ�ѧ����ʽΪ2H2O2$\frac{\underline{\;MnO_2\;}}{\;}$2H2O+O2����װ��D�з�����Ӧ�Ļ�ѧ����ʽΪSO2+2NaOH=Na2SO3+H2O��
���� ��1��װ��B��ҩƷ��Ũ���ᣬŨ���������ˮ�ԣ����ȼ�����ɶ����������壬��������������������������Һ��Ӧ�����������ƺ�ˮ���ݴ˽��з������
��2�����������ڶ������̵Ĵ�����������ˮ��������д����Ӧ�Ļ�ѧ����ʽ���ɣ�
��� �⣺��1��װ��B��ҩƷ��Ũ���ᣬŨ���������ˮ�ԣ�װ��B�������dz�ȥˮ�������������������ˮ�֣���
װ��C�����ȼ�յ������������ɫ���棬���ȼ�����ɶ����������壬��������������������������Һ��Ӧ�����������ƺ�ˮ����װ��D�����������ն�������ֹ��Ⱦ������
��2�����������ڶ������̵Ĵ�����������ˮ����������Ӧ�Ļ�ѧ����ʽΪ��2H2O2$\frac{\underline{\;MnO_2\;}}{\;}$2H2O+O2����
װ��D�з�����Ӧ�Ļ�ѧ����ʽΪSO2+2NaOH=Na2SO3+H2O��
�ʴ�Ϊ����1����ȥˮ�������������������ˮ�֣�����������ɫ���棻���ն�������ֹ��Ⱦ������
��2��2H2O2$\frac{\underline{\;MnO_2\;}}{\;}$2H2O+O2����SO2+2NaOH=Na2SO3+H2O��
���� �����ѶȲ������п�����Ҫ����֮һ����������ʵ��������ȡ�����ķ�Ӧԭ���������Ļ�ѧ���ʵ�����ȷ�����Ĺؼ���

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д���1��2����ԭ��2P��
��2�����������NO3-��
��3�������к������Ľ���Ԫ��Ca��
��4������He��
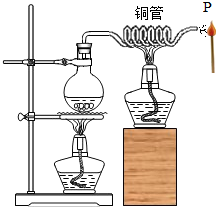 ��ͼ��ʾ��������ƿ�е�ˮʹ֮���ڣ�ˮ������ͭ��������ѻ���ܿ�P��������ȣ�Ѹ���ƿ�����ʼȼ�գ�����˵��������ǣ�������
��ͼ��ʾ��������ƿ�е�ˮʹ֮���ڣ�ˮ������ͭ��������ѻ���ܿ�P��������ȣ�Ѹ���ƿ�����ʼȼ�գ�����˵��������ǣ�������| A�� | ˮ���ڣ�ˮ����������������� | |
| B�� | P��ˮ������������ʹ����¶ȴﵽ�Ż�� | |
| C�� | �������ˮ��������������ֽӴ����Ӷ�ȼ�� | |
| D�� | ���ȼ�յIJ��ﺬ�ж�����̼��ˮ |
| ѡ�� | ��� | ���� |
| A | �Ͻ� | �֡����������� |
| B | �� | ʳ�Ρ�����ʯ�� |
| C | �л��� | ���顢�Ҵ��������� |
| D | ��ʴ������ | Ũ���ᡢ�������� |
| A�� | A | B�� | B | C�� | C | D�� | D |
| A�� | ��Լ��ˮ�ͷ���ˮ����Ⱦ�ǰ���ˮ��Դ����Ч;�� | |
| B�� | ˮ����������������ɵĻ���� | |
| C�� | ����ˮ����ˮ�����������õķ������������������������� | |
| D�� | �������ô���ˮ�������彡�� |
| A�� |  ȡ���Ȼ��ƹ��� | B�� |  �����Ȼ��ƹ��� | C�� | 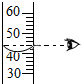 ��ȡˮ����� | D�� |  �ܽ� |
 ��1�����ǵؿ��к������Ľ���Ԫ�أ���Ԫ�����ڱ�����Ԫ�ص���Ϣ��ͼ��ʾ��
��1�����ǵؿ��к������Ľ���Ԫ�أ���Ԫ�����ڱ�����Ԫ�ص���Ϣ��ͼ��ʾ��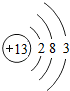 ��
�� ��ѧ������ϢϢ��أ�����ѧ���Ļ�ѧ֪ʶ����������⣺
��ѧ������ϢϢ��أ�����ѧ���Ļ�ѧ֪ʶ����������⣺