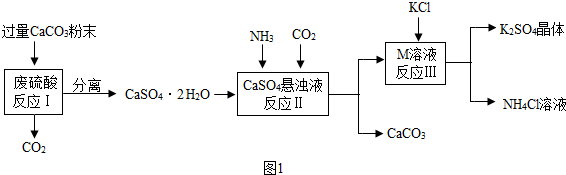
��Ŀ����
15��2015��4��25���Ჴ�����ӵ�����������8.1���������ɴ�������Ա�����ͲƲ���ʧ���й�������ʱ�ɳ���Ԯ��ǰ������Ϊ�Ჴ�������ṩ�˴���������Һ�������������С�84������Һ�������������ߣ�����Ҫ�ɷ��Ǵ������ƣ�NaClO������ȡ���Ļ�ѧ����ʽΪ2NaOH+X�TNaClO+NaCl+H2O����X�Ļ�ѧʽΪCl2��NaClO����Ԫ�صĻ��ϼ�Ϊ+1�������к��е���Ҫ�ɷ�Ϊ���࣮���� �������غ㶨�ɣ���Ӧǰ��ԭ�����ࡢ��Ŀ�����䣬�ݴ��ɷ�Ӧ�Ļ�ѧ����ʽ�ƶϷ�Ӧ��X�Ļ�ѧʽ��
�����ڻ��������������ϼ۴�����Ϊ�㣬���NaClO�Ļ�ѧʽ���н��
����������������Ӫ���ص����ࡢʳ����Դ���������������ʳ���ж�������Ӫ���أ����з������
��� �⣺�������غ㶨�ɣ���Ӧǰ��ԭ�����ࡢ��Ŀ�����䣬�ɷ�Ӧ�Ļ�ѧ����ʽ2NaOH+X�TNaClO+NaCl+H2O����Ӧǰ�ơ�������ԭ�Ӹ����ֱ�Ϊ2��2��2����Ӧ������������ơ������⡢��ԭ�Ӹ����ֱ�Ϊ2��2��2��2�����ݷ�Ӧǰ��ԭ�����ࡢ��Ŀ���䣬��ÿ��X������2����ԭ�ӹ��ɣ�������X�Ļ�ѧʽΪCl2��
��Ԫ����+1�ۣ���Ԫ����-2�ۣ�����Ԫ�صĻ��ϼ���x�������ڻ��������������ϼ۴�����Ϊ�㣬�ɵã���+1��+x+��-2��=0����x=+1�ۣ�
�����и������ۣ������������࣮
�ʴ�Ϊ��Cl2��+1�����࣮
���� �����ѶȲ������û�ѧ��Ӧǰ��ԭ���غ���ȷ�����ʵĻ�ѧʽ�����������������ϼ۴�����Ϊ�㡢����Ӫ���ص�����������ʳ����Դ������ȷ��������Ĺؼ���

��ϰ��ϵ�д�
�����Ŀ
11������������������ǣ�������
| A�� | H2SO4 | B�� | SO2 | C�� | KOH | D�� | O2 |
12������ȥ���������е��������ʣ�������Ϊ���ʣ�������ȡ�ķ�����ȷ���ǣ�������
| ѡ�� | ���� | ������Լ������ |
| A | NaCl ��Һ��CaCl2�� | ������̼������Һ������ |
| B | CaCO3��CaO�� | �{������ |
| C | Cu��NO3��2 ��Һ��AgN03�� | ����ͭ�ۣ����� |
| D | CO2��HC1�� | ����������������Һ��ϴ�� |
| A�� | A | B�� | B | C�� | C | D�� | D |
10����3.52g CO2��SO2�������ͨ��һ������NaOH��Һ�У�����ȫ����NaOH��Ӧ�����Σ�����Ӧ�����Һ��ѹ�������ɺ�õ������ᾧˮ�Ĺ��壬�������������Ϊ��������
| A�� | 8.48 g | B�� | 7.48 g | C�� | 5.72 g | D�� | 5.22g |
20�� ������ϡ������μ��뵽12.5gˮ����ĩ�У��������Ķ�����̼���建��ͨ�뵽������NaOH��Һ�У����6������NaOH��Һ��������������������ʾ���£�
������ϡ������μ��뵽12.5gˮ����ĩ�У��������Ķ�����̼���建��ͨ�뵽������NaOH��Һ�У����6������NaOH��Һ��������������������ʾ���£�
��1�����������ݿ��Է����ó��������ۣ�
��ʵ���й�����4.4g CO2���壻
����ʱ����Ƴ٣�ϡ������ˮ���ķ�Ӧ���ʼ�С���������С�������䡱������������ǵ�λʱ��������������Һ���ӵ�����Խ��ԽС��
��2����ˮ����̼��Ƶ���������80%����Է���������CaCO3-100��CO2-44
��3��ʵ���в��̼��Ƶ���������ƫ����ܵ�ԭ������������Һ�����˿����еĶ�����̼����������ˮ���������Ȼ���ȣ�
��4����ˮ����ѧʽ�ɱ�ʾΪMg��OH��2•2CaCO3��д����ˮ����ϡ�����ܷ�Ӧ����ʽ��Mg��OH��2•2CaCO3+6HCl�T2CaCl2+MgCl2+4H2O+2CO2����
 ������ϡ������μ��뵽12.5gˮ����ĩ�У��������Ķ�����̼���建��ͨ�뵽������NaOH��Һ�У����6������NaOH��Һ��������������������ʾ���£�
������ϡ������μ��뵽12.5gˮ����ĩ�У��������Ķ�����̼���建��ͨ�뵽������NaOH��Һ�У����6������NaOH��Һ��������������������ʾ���£�| ʱ��/min | 0 | 1 | 2 | 3 | 4 | 5 | 6 |
| ���ӵ�����/g | 0 | 2.2 | 3.2 | 3.8 | 4.2 | 4.4 | 4.4 |
��ʵ���й�����4.4g CO2���壻
����ʱ����Ƴ٣�ϡ������ˮ���ķ�Ӧ���ʼ�С���������С�������䡱������������ǵ�λʱ��������������Һ���ӵ�����Խ��ԽС��
��2����ˮ����̼��Ƶ���������80%����Է���������CaCO3-100��CO2-44
��3��ʵ���в��̼��Ƶ���������ƫ����ܵ�ԭ������������Һ�����˿����еĶ�����̼����������ˮ���������Ȼ���ȣ�
��4����ˮ����ѧʽ�ɱ�ʾΪMg��OH��2•2CaCO3��д����ˮ����ϡ�����ܷ�Ӧ����ʽ��Mg��OH��2•2CaCO3+6HCl�T2CaCl2+MgCl2+4H2O+2CO2����
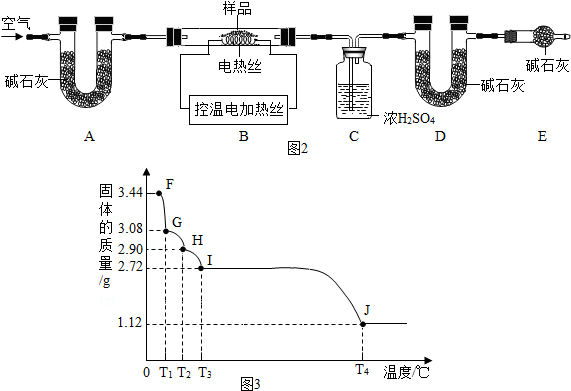
4������ͼʾ��ʵ������У���ȷ���ǣ�������
| A�� |  �ιܵ�ʹ�� | B�� |  ���������� | ||
| C�� |  ���� | D�� |  ������������ |
5����ѧ��ȤС���ͬѧ��������Ҫ�ɷ���ʯ��������ȼ�ս���������̽����
��1��ȡһ֧��ɫ�Ķ�������С������һС�飬��������ˮ�У�����һֱ����ˮ���ϣ��ݴ����ܽ������������������������ʣ�Ӳ��С��������ˮ���ܶ�С��ˮ��
��2����ȼ����ͼһ�����ܹ۲쵽������ѧ�仯�������⣬���ȣ�ȼ�գ���д��һ����
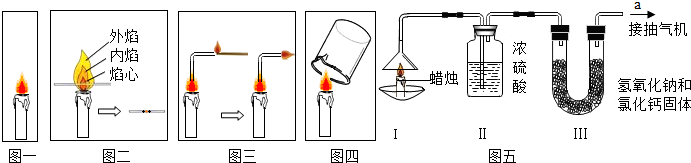
��3��ȡһСľ����Ѹ��ƽ��������У�ͼ����Լһ���ȡ�����۲쵽ľ��������ֻ��ν��ߣ��ݴ��Ʋ⣬��������������¶���ߣ�
��4�����̲����ܲ������ģ���ȼ�յ�ľ������������һ�ˣ�ͼ���������ֵ�����������Ե�ȼ�������Ʋ����������ܻ���ʯ��������
��5��ȡһֻ��������С�ձ�������������Ϸ���ͼ�ģ����۲��ձ�������ˮ����֣�Ƭ�̺�ȡ���ձ���Ѹ�����ձ��е������������ʯ��ˮ�����۲쵽ʯ��ˮ����ǣ�
�ݴ�С���ó���������̼����Ԫ����ɵģ�����Ϊ���Ľ����Ƿ���ȷ������ȷ��
��6��Ϊ�˲ⶨ������̼������ ��Ԫ�ص������ȣ�С���������ͼ����ʾ��ʵ�飮
ʵ�鲽�����£�
�ȷֱ��������װ�â�����ˮ����װ�â����ն�����̼����������
��ͼʾ���Ӻ���������ȼ����ͬʱ��a���ܿڳ�����
һ��ʱ���Ϩ�������ٷֱ��������װ�â�װ�â��������
ʵ�������������ش�
�ٸ�ʵ���в��ˮ������Ϊ1.8g��
���ɸ�ʵ�����ݼ��㣬������̼����Ԫ�ص�����֮��Ϊ6��1��
�������ϣ�װ�â��װ�â����ӵ���������������ʧȥ����������ԭ���������ĵ�����Ƚϣ����ɵĶ�����̼��ˮ�������˲μӷ�Ӧ������������Ԫ��������
�ܸ�ʵ���ܷ�ȷ�������ȼ�����ɶ�����̼��ˮ��������˵�����ɣ����ܣ������е�ˮ�Ͷ�����̼Ҳ�ܱ�װ�â�װ�â����գ�
��1��ȡһ֧��ɫ�Ķ�������С������һС�飬��������ˮ�У�����һֱ����ˮ���ϣ��ݴ����ܽ������������������������ʣ�Ӳ��С��������ˮ���ܶ�С��ˮ��
��2����ȼ����ͼһ�����ܹ۲쵽������ѧ�仯�������⣬���ȣ�ȼ�գ���д��һ����

��3��ȡһСľ����Ѹ��ƽ��������У�ͼ����Լһ���ȡ�����۲쵽ľ��������ֻ��ν��ߣ��ݴ��Ʋ⣬��������������¶���ߣ�
��4�����̲����ܲ������ģ���ȼ�յ�ľ������������һ�ˣ�ͼ���������ֵ�����������Ե�ȼ�������Ʋ����������ܻ���ʯ��������
��5��ȡһֻ��������С�ձ�������������Ϸ���ͼ�ģ����۲��ձ�������ˮ����֣�Ƭ�̺�ȡ���ձ���Ѹ�����ձ��е������������ʯ��ˮ�����۲쵽ʯ��ˮ����ǣ�
�ݴ�С���ó���������̼����Ԫ����ɵģ�����Ϊ���Ľ����Ƿ���ȷ������ȷ��
��6��Ϊ�˲ⶨ������̼������ ��Ԫ�ص������ȣ�С���������ͼ����ʾ��ʵ�飮
ʵ�鲽�����£�
�ȷֱ��������װ�â�����ˮ����װ�â����ն�����̼����������
��ͼʾ���Ӻ���������ȼ����ͬʱ��a���ܿڳ�����
һ��ʱ���Ϩ�������ٷֱ��������װ�â�װ�â��������
ʵ�������������ش�
| ���� | װ�â� | װ�â� | |
| ��Ӧǰ������/g | 15.8 | 182.3 | 212.2 |
| ��Ӧ�������/g | 14.4 | 184.1 | 216.6 |
���ɸ�ʵ�����ݼ��㣬������̼����Ԫ�ص�����֮��Ϊ6��1��
�������ϣ�װ�â��װ�â����ӵ���������������ʧȥ����������ԭ���������ĵ�����Ƚϣ����ɵĶ�����̼��ˮ�������˲μӷ�Ӧ������������Ԫ��������
�ܸ�ʵ���ܷ�ȷ�������ȼ�����ɶ�����̼��ˮ��������˵�����ɣ����ܣ������е�ˮ�Ͷ�����̼Ҳ�ܱ�װ�â�װ�â����գ�