��Ŀ����
ij���ʰ�װ���ϵIJ���˵����ͼ��ʾ����1����������ڻ������е� ������ţ���
A���� B���� C���� D��������
��2�������Ӧ���е������� ������ţ���
A��������ˮ B���лӷ��� C�������ֽ�
��3������淋���Է������� ����Ԫ�ص���������Ϊ ��
��4�����������ǿ�Ӧ���������ռӦ�ķ���ʽΪ��NH4NO3+NaOH=NaNO3+NH3��+X����X�Ļ�ѧʽ�� ��ʩ�ø������ʱ��Ҫ������ ����ᡱ��������ʻ��ã��������ͷ�Ч��

���𰸡���������1��笠����Ӻ����������ɵĻ����������Σ�
��2����������淋����淽�������ж������ʣ�
��3���������ʵĻ�ѧʽ���Լ�����Է���������Ԫ�ص�����������
��4�����ݻ�ѧ����ʽ�����ж����ʵĻ�ѧʽ��
����⣺��1�����������笠����Ӻ������������ɵĻ���������Σ����C��
��2�����������ʱ��Ӧ�÷�������ɹ��˵�������������ˮ�����ȶ��Բ���AC��
��3������淋���Է�������Ϊ��14×2+1×4+16×3=80����Ԫ�ص���������Ϊ��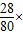 100%=35%�����80��35%��
100%=35%�����80��35%��
��4����NH4NO3+NaOH=NaNO3+NH3��+X��֪��ÿ��X�к���1����ԭ�Ӻ�2����ԭ�ӣ���ˮ�����H2O��
�̬���ʺ��Լ������ʻ�������ɰ������Ӷ����ͷ�Ч������
������������Ҫ�������ʵķ��ࡢ���ʡ����ݻ�ѧʽ����ȷ����֪ʶ������Ԫ����������ʱ��ע�ⲻҪ©���ٷֺţ�
��2����������淋����淽�������ж������ʣ�
��3���������ʵĻ�ѧʽ���Լ�����Է���������Ԫ�ص�����������
��4�����ݻ�ѧ����ʽ�����ж����ʵĻ�ѧʽ��
����⣺��1�����������笠����Ӻ������������ɵĻ���������Σ����C��
��2�����������ʱ��Ӧ�÷�������ɹ��˵�������������ˮ�����ȶ��Բ���AC��
��3������淋���Է�������Ϊ��14×2+1×4+16×3=80����Ԫ�ص���������Ϊ��
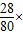 100%=35%�����80��35%��
100%=35%�����80��35%����4����NH4NO3+NaOH=NaNO3+NH3��+X��֪��ÿ��X�к���1����ԭ�Ӻ�2����ԭ�ӣ���ˮ�����H2O��
�̬���ʺ��Լ������ʻ�������ɰ������Ӷ����ͷ�Ч������
������������Ҫ�������ʵķ��ࡢ���ʡ����ݻ�ѧʽ����ȷ����֪ʶ������Ԫ����������ʱ��ע�ⲻҪ©���ٷֺţ�

��ϰ��ϵ�д�
�����Ŀ
 ��2007?��ۣ�ij���ʰ�װ���ϵIJ���˵����ͼ��ʾ��
��2007?��ۣ�ij���ʰ�װ���ϵIJ���˵����ͼ��ʾ�� ij���ʰ�װ���ϵIJ���˵����ͼ��ʾ��
ij���ʰ�װ���ϵIJ���˵����ͼ��ʾ�� 17��ij���ʰ�װ���ϵIJ���˵����ͼ��ʾ��
17��ij���ʰ�װ���ϵIJ���˵����ͼ��ʾ�� 13��ij���ʰ�װ���ϵIJ���˵����ͼ��ʾ������������и��⣺
13��ij���ʰ�װ���ϵIJ���˵����ͼ��ʾ������������и��⣺