��Ŀ����
��κ�������Ч����Լ��ʹ��ú��ʯ�͡���Ȼ����������������Ҫ�Ļ�ʯȼ�ϣ��ӽ��������������������Դ��ȫ�������ٵĹ�ͬ���⣬�Ƿ�չ��̼���õĵ���֮�����ش��������⣺
������1����Ȼ����̫ԭ�м�ͥ����Ҫ����ȼ�ϡ�Ϊ��ֹ
�� ��Ȼ��й©���Σ�գ����ڼ��а�װ���������ڼס���
��Ȼ��й©���Σ�գ����ڼ��а�װ���������ڼס���
��ͼ�У���װλ����ȷ���� ����ס����ҡ�����
д����Ȼ��ȼ�յĻ�ѧ����ʽ�� ��
������2����ֱ����ú������ȼ�ϼ���Ⱦ�������˷���Դ��úȼ�� ʱ������
ʱ������
���γ��������Ҫ�� ��֮һ��ú�������ǰ�ú��Ϊ�����Դ����Ҫһ��������һ����Ҫ��Ӧ�ǣ���ˮ����ͨ�����ȵ�ú�㣬�õ�һ����̼���������÷�Ӧ�Ļ�ѧ����ʽΪ ��
��֮һ��ú�������ǰ�ú��Ϊ�����Դ����Ҫһ��������һ����Ҫ��Ӧ�ǣ���ˮ����ͨ�����ȵ�ú�㣬�õ�һ����̼���������÷�Ӧ�Ļ�ѧ����ʽΪ ��
������3���ҹ������з���һ�ֵ綯����ʹ�õ���Դ������﮵�أ��ӻ�ѧ�Ƕȿ�����ʹ�����͵�����������ŵ���
�����ٳ����㼴�ɣ�
�� ��1���� CH4+2O2 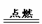 C
C O2+2H2O ��2��SO2����������� C+H2O====CO+H2
O2+2H2O ��2��SO2����������� C+H2O====CO+H2
��3������Ⱦ�����������̼�ŷŻ��Լ��ʯ��Դ�ȣ����������𰸾��ɣ�

С�־�����ܴ�������������ʦ���ѣ�ѧ�ꡰ��Ļ�ѧ���ʡ����������������ۡ�ʳ���ڳ����е����á���
(1)��ϰ��Ļ�ѧ����
| a.����ָʾ������ | ����Һ��ʹ��ɫʯ����Һ���ɫ | ����Һ����ʹ��ɫ��̪��Һ��ɫ |
| b. | Fe��2HCl===FeCl2��H2�� | Fe��H2SO4===FeSO4��H2�� |
| c.����ij�������������� | Fe2O3��HCl===FeCl3��H2O | ���� |
| d.���� |
��������С�ֵĿ��ñʼ�û���ã���c.����ijЩ�������������á�����Ļ�ѧ����ʽδ��ƽ�������д����ƽ��Ļ�ѧ����ʽ��_______________��
(2)���۽���
�� С���Ժ���˵��Ŀǰ����������֯�����ƹ�ʹ���й�������ʹ����������ʱ���ŵ�ʳ�ף����Բ���������Ҫ����Ԫ��__________Ԫ�أ��÷�Ӧ��ԭ������������Ļ�ѧ����___________(����š�a����b����c��)��
�� �����С�֣�ʳ������ԶԶ��ֹ��Щ�أ���_____________ (��дһ��ʳ���������е�����)��
(3)��˼
���ᡢ���ᡢʳ�������ƻ�ѧ���ʵ�ԭ������Ϊ���ǵ�ˮ��Һ�ж�����__________���ӡ�
ij��ȤС���ͬѧ��������װ����ʵ������ȡ������̼���壬��ش��������⣺
|

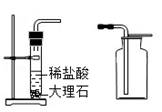

A B C
��2��ͼ
��1��ͼ������a�������� ��
��2���Ʊ�����֮ǰ��������Ҫ���װ�õ������ԣ���д�����Aװ�������Եķ�����
��
��3��Aװ�ó�����ʵ������ȡ������̼�⣬����������ȡ ���ѧʽ�����壬��Ӧ�Ļ�ѧ����ʽΪ ��
��4����Bװ���ռ��ռ�������̼ʱ����μ��������̼�Ƿ��ռ�����
��
��5��Cװ���ǽ�ϸͭ˿���С������Թܵײ����Թܵײ����𣩣����ϴ�������Ƥ���ĵ��ܺ��ձ���װ����һ����ʵ������ȡ�����װ�ã��ɽ�Aװ�øij�Cװ������ʵ������ȡ������̼����һ�Ľ�����ҪĿ���� ��ѡ����ţ���
A��ʹ��Ӧ��ʱ������ֹͣ B��ʹ������Թܵõ����� C�����ɸ����CO2
 ������ý�������
������ý������� A�������Գ�ȥˮ�еIJ��������ʡ�ɫ�ء����ֳ�ζ
A�������Գ�ȥˮ�еIJ��������ʡ�ɫ�ء����ֳ�ζ

