��Ŀ����
������������������֮������ϵ���ǻ�ѧѧϰ��һ����Ч��������ͼ��ʾ��������A-H���Ĺ�ϵ����������ʾ���ʼ��ת����ϵ����--����ʾ���˵������ܷ�����ѧ��Ӧ����A��G���ǵ��ʣ�����C���������F��һ����ɫ��Һ������������ũҩ������Һ��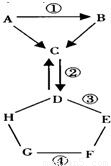
����ش�
��1��C�Ļ�ѧʽ ��H������ ��
��2��д����Ţ١��ڡ��ۡ��ܵĻ�ѧ����ʽ��
�� ��
�� ��
�� ��
�� ��
����⣺��1������C��������𣬿�֪CΪ������̼����ôD����̼���Σ�����H��E������ͼ�ָ���FΪ����ͭ������֪��EΪ����Կ���֪��HΪ�ᣬ���������ƶϳ�DΪ̼���ƣ���E�����������ƣ��ʱ����Ϊ��CO2 HCl
��2������CΪ������̼������֪��AΪ��������̼���ʣ�CΪһ����̼��FΪ����ͭ��HΪ�ᣬ�����ܹ�����������Gֻ���ǻ��õĽ�������������ͬʱ��ϣ�1���Ľ�����������ĸ���ѧ����ʽ���ʱ����Ϊ��
��2C+O2
 2CO
2CO��2NaOH+CO2=Na2CO3+H2O
��Na2CO3+Ca��OH��2=CaCO3��+2NaOH
��Fe+CuSO4=Cu+FeSO4
�������������ʵ��ƶϷ���������ͻ�ƿڵ�ǰ���¸���ͼ�����ƻ����ƣ����߽��Ҳ�ɣ��Ʋ���������Ŀ����֤���Ƿ�ÿһ�������ʣ������е�A�Ʋ�Ϊ��������̼���ʶ���Ӱ�챾��Ľ��ͬʱ���ո�����Ŀ�е���Ϣ����ȷ��д��ѧ����ʽ��

ѧ����������������ѧϰ����Ҫ�ֶΡ����ж��й�����֪ʶ�Ĺ�����ȫ��ȷ��һ����
| A�� ������; | B�� Ԫ��֮�� |
| Һ̬�������������ȼ�� ������̼�������������� | �����к�������Ԫ�ة�����Ԫ�� �ؿ��� |
| C�����彡�� | D�������� |
| ȱ�Ʃ������������� ȱ��������ƶѪ�� | ����������������С���¶ȸı� ʪ�·����ɩ��������м�϶ |
ѧ����������������ѧϰ����Ҫ�ֶΡ����ж��й�����֪ʶ�Ĺ�����ȫ��ȷ��һ����
| A�� ������; | B�� Ԫ��֮�� |
| Һ̬�������������ȼ�� ������̼�������������� | �����к�������Ԫ�ة�����Ԫ�� �ؿ��к������Ľ���Ԫ�ة�����Ԫ�� |
| C�����彡�� | D�������� |
| ȱ�Ʃ������������� ȱ��������ƶѪ�� | ����������������С���¶ȸı� ʪ�·����ɩ��������м�϶ |
ѧ����������������ѧϰ����Ҫ�ֶΡ����ж��й�����֪ʶ�Ĺ�����ȫ��ȷ��һ����
| A�� ������; | B�� Ԫ��֮�� |
| Һ̬�������������ȼ�� ������̼�������������� | �����к�������Ԫ�ة�����Ԫ�� �ؿ��к������Ľ���Ԫ�ة�����Ԫ�� |
| C�����彡�� | D�������� |
| ȱ�Ʃ������������� ȱ��������ƶѪ�� | ����������������С���¶ȸı� ʪ�·����ɩ��������м�϶ |
 �������Ľ���Ԫ�ة�����Ԫ��
�������Ľ���Ԫ�ة�����Ԫ��