��Ŀ����
7�� ��̼��������ḻ��ʣ�������һ���߽���̼�������磮
��̼��������ḻ��ʣ�������һ���߽���̼�������磮��1�����ݱ����ṩ����Ϣ���ش����⣮
| ���ʯ | ʯī | C60 | |
| �ṹ |  |  |  |
| ��; | �ò������и����ʯ�����������ͷ | ���缫���� | ����������� |
| ��Ӧ������ | Ӳ�ȴ� | �����ĵ����� |  |
��2����ͼ��ʾ�ġ�̼���ࡱ����֪����Ĺ�����ϣ�����̼Ԫ����ɣ����ж�ṹ�����Ժã���ʯ���к�ǿ����������������ˮ�����������ʯ�ͼ������Կɻָ�ԭ״�����й��ڡ�̼���ࡱ��˵������ȷ����B������ţ�
A���ɴ�������ʯ��й© B�������ظ�ʹ��
C����һ�������¿ɻ�ԭ����ͭ D������������ȫȼ�յIJ�����CO2
��3��������Ⱦ��ս���ǽ������绷���յ��й����⣮�������ŷŵ�β���г�����NO��CO�����壮�����������װһ��β������װ�ã��ڴ����������£�ʹNO��CO������Ӧ���������ֲ������ѭ�������壬д���÷�Ӧ�Ļ�ѧ����ʽ2NO+2CO$\frac{\underline{\;����\;}}{\;}$N2+2CO2��
���� ��1�����ݽ��ʯ��������������״��Ӳ�ȴ�ʯī���������ĵ����ԵȽ��з������
��2���������⣬��̼���ࡱ����֪����Ĺ�����ϣ�����Ҫ�ɷ���ʯīϩ��̼���ܣ����߶���̼���ʣ����߱��ߵ��Ժ����ɶ�Ľṹ���������ʯ�ͼ������Կɻָ�ԭ״���ݴ˽��̼���ʵ����ʽ��з����жϣ�
��3�����ݷ�Ӧ��������Ӧ�������������غ㶨�ɿ�����д��ѧ����ʽ��
��� �⣺��1�����ʯ��������������״��Ӳ�ȴ�������̽����ͷ���и���ȣ�ʯī���缫���ϣ���������ʯī���������ĵ����ԣ����ʯ��ʯī�������ʲ���ܴ�����Ϊ���ǵĽṹ��̼ԭ�ӵ����з�ʽ��ͬ��
��2��A�������⣬��̼���ࡱ�������ɶ�Ľṹ�����������ԣ����Կɴ�������ʯ��й©����ѡ��˵����ȷ��
B����̼���ࡱ�������ʯ�ͼ������Կɻָ�ԭ״�����Կ��ظ�ʹ�ã���ѡ��˵������
C����̼���ࡱ��Ҫ�ɷ���ʯīϩ��̼���ܣ����߶���̼���ʣ������л�ԭ�ԣ���һ�������¿ɻ�ԭ����ͭ����ѡ��˵����ȷ��
C����̼���ࡱ��Ҫ�ɷ���ʯīϩ��̼���ܣ����߶���̼���ʣ��������»�ѧ���ʲ����ã���ѡ��˵������
D����̼���ࡱ��Ҫ�ɷ���ʯīϩ��̼���ܣ����߶���̼���ʣ�������������ȫȼ�յIJ�����CO2����ѡ��˵����ȷ��
��3��CO��NO������Ӧ��������������Ⱦ�����壬����һ��Ϊ������̼������һ���ǿ���������������嵪������Ӧ�Ļ�ѧ����ʽΪ��2NO+2CO$\frac{\underline{\;����\;}}{\;}$N2+2CO2��
�ʴ�Ϊ����1��
| ���ʯ | ʯī | C60 | |
| �ṹ | |||
| ��; | �ò������и����ʯ�����������ͷ | ||
| ��Ӧ������ | �����ĵ����� |
���� �����ѶȲ�������̼���ʵ��������ʡ���ѧ��������;������ʽ����д����ȷ����Ĺؼ���

 ͬ��������ϰϵ�д�
ͬ��������ϰϵ�д� �ο�ͨ�γ̱�˼ά����������ѵ��ϵ�д�
�ο�ͨ�γ̱�˼ά����������ѵ��ϵ�д�| A�� | CO2��һ��������̼�����к���������ԭ�� | |
| B�� | 2H��������Ԫ�� | |
| C�� | Mg+2��һ��þ���Ӵ���������λ����� | |
| D�� | S2-����Ԫ�صĻ��ϼ�Ϊ������ |
| ѡ�� | ���� | �������� |
| A | CaO��ĩ��CaCO3��ĩ�� | ��������ˮ����ֽ������� |
| B | Fe��Fe2O3�� | �������ϡ���ᣬ��ַ�Ӧ����� |
| C | NaOH��Һ[Ca��OH��2��Һ] | ͨ�����CO2���壬��ַ�Ӧ����� |
| D | CuSO4��Һ�����ᣩ | �������CuO��ĩ�����ȣ���ַ�Ӧ����� |
| A�� | A | B�� | B | C�� | C | D�� | D |
| A�� | ���������ྻ��Դ | B�� | ȫ���ͣ����Ⱦ�Ļ�����ҵ | ||
| C�� | ����ʹ�ù�����ͨ���� | D�� | ���ع��ͱ��������� |
| A��ѧ������ | B��ѧ�밲ȫ |
| �ٵ�������·��ϣ����þƾ���ϴ��ȥ �������н��ʯ�IJ������ò��� �������������Ƶ��� | �ٳ��˹��Ż�����ù��Ǹǻ� ��������һ��ˮ�ܷ�ֹú���ж� ��ҹ�䷢��Һ����й©����Ѱ��й©Դ |
| C��ѧ����Դ | D��ѧ�뷢�� |
| �ٷ�ֹ������ʴ�DZ���������Դ��;��֮һ ��ú��ʯ�͡���Ȼ���ǿ�������Դ �ۺ������̲��ŷḻ�Ļ�ѧ��Դ | �ٲ�����������ָʾ�� ���Ž��з���Ԫ�������ɲ�����Ԫ�����ڱ� �ۺ�°��Ƽָ������ȡ���� |
| A�� | A | B�� | B | C�� | C | D�� | D |
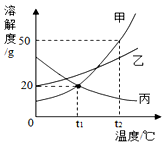
| A�� | t1��ʱ���ױ�����������Һ����������������� | |
| B�� | ��t1��ʱ�������ʵı�����Һ������t2��ʱ��ֻ�б����������� | |
| C�� | t2��ʱ����30g�����ʼ��뵽50gˮ�в��Ͻ������ܽ⣬�γɵ���Һ��������80g | |
| D�� | t2��ʱ�����ס��ҵı�����Һ��������10gˮ�������������������һ���� |
| ѡ�� | ʵ��Ŀ�� | ʵ�鷽�� |
| A | �����Ȼ��ƺ������ | ��ˮ�ܽ⡢�����¶� |
| B | ����CO2���Ƿ��е�����CO | ��ȼ���壬�۲��ܷ�ȼ�� |
| C | ����������������̼�͵��� | �������ʯ��ˮ���۲����ʯ��ˮ�ı仯 |
| D | ������ë�Ͳ�˿ | ��ȼ������ζ |
| A�� | A | B�� | B | C�� | C | D�� | D |