��Ŀ����
20��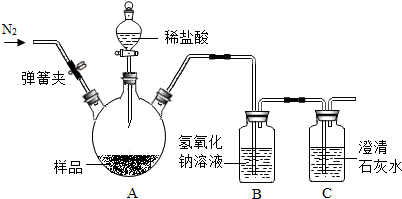 ͬѧ���ڰ�����ʦ����ʵ����ʱ�����ְ�ƿ���õ��������Ʒ�ĩ[Ca��OH��2]����Ҿ�����ʵ��ķ���̽����ƿ��ĩ����ɣ������ϣ�����ʱ��Ca��OH��2���ܽ����0.165g��
ͬѧ���ڰ�����ʦ����ʵ����ʱ�����ְ�ƿ���õ��������Ʒ�ĩ[Ca��OH��2]����Ҿ�����ʵ��ķ���̽����ƿ��ĩ����ɣ������ϣ�����ʱ��Ca��OH��2���ܽ����0.165g��ʵ��һ����Ʒ����ˮ
ȡһҩ����Ʒ������20mLˮ�г���ܽ���г����
��1����ͬѧ��Ϊ���ó�����������������ƣ����ƶϵ�����Ӧ��������ʱ��Ca��OH��2���ܽ�Ⱥ�С��
��2��Ҫ֤����Ʒ�Ѿ���������̼��ƣ�����������ij������еμ�ϡ���ᣬ��Ӧ�Ļ�ѧ����ʽΪCaCO3+2HCl�TCaCl2+H2O+CO2����
ʵ������ⶨ��Ʒ��̼��Ƶ���������
��ͼ�����ɼУ���ͨ��N2һ��������ž�װ���к��е�CO2���رյ��ɼУ���A����Ʒ�м���������ϡ���ᣮͨ���ⶨ��Ӧ�����Ķ�����̼������������ɼ���̼��Ƶ�����������װ���������Ѽ��飬�г�װ����ʡ�ԣ���
��3��װ��B�з�����Ӧ�Ļ�ѧ����ʽ��2NaOH+CO2�TNa2CO3+H2O��
��4��ʵ�������Ҫ��ͨһ���N2��Ŀ����ʹ��Ӧ�����Ķ�����̼ȫ��������������Һ���գ�
��5����C���ޱ仯�����ⶨBƿ����m g��Ҫ������Ʒ��̼��Ƶ�������������ҪԤ�Ȳⶨ��ƷA��������
��6����C��ʯ��ˮ����ǣ����õ���Ʒ��CaCO3 ����������ֵ��ƫ�ͣ��ƫ�ߡ�����ƫ�͡����䡱����
���� ����ʱ�������Ƶ��ܽ�Ⱥ�С��
̼��ƺ�ϡ���ᷴӦ�����Ȼ��ơ�ˮ�Ͷ�����̼��
������̼�ܺ��������Ʒ�Ӧ����̼���ƺ�ˮ��
B�����ӵ�������Ϊ��Ӧ���ɵĶ�����̼�����������ݶ�����̼���������Լ���̼��Ƶ���������һ�����Լ�����Ʒ��̼��Ƶ�����������
��� �⣺��1���ó�����������������ƣ����ƶϵ�����Ӧ���ǣ�����ʱ��Ca��OH��2���ܽ�Ⱥ�С��һҩ���������Ʋ���ȫ���ܽ���20mLˮ�У�
�������ʱ��Ca��OH��2���ܽ�Ⱥ�С��
��2��̼��ƺ�ϡ���ᷴӦ�Ļ�ѧ����ʽΪ��CaCO3+2HCl�TCaCl2+H2O+CO2����
���CaCO3+2HCl�TCaCl2+H2O+CO2����
��3��װ��B�ж�����̼���������Ʒ�����Ӧ�Ļ�ѧ����ʽΪ��2NaOH+CO2�TNa2CO3+H2O��
���2NaOH+CO2�TNa2CO3+H2O��
��4��ʵ�������Ҫ��ͨһ���N2��Ŀ����ʹ��Ӧ�����Ķ�����̼ȫ��������������Һ���գ�
���ʹ��Ӧ�����Ķ�����̼ȫ��������������Һ���գ�
��5��Ҫ������Ʒ��̼��Ƶ�������������ҪԤ�Ȳⶨ��ƷA��������
�����ƷA��
��6����C��ʯ��ˮ����ǣ�˵���ж�����̼û�б�����������Һ���գ��ᵼ�¼������̼�������ƫС���Ӷ��������õ���Ʒ��CaCO3 ����������ƫ�ͣ�
���ƫ�ͣ�
���� �������ʵ�飬��ѧ�ؽ���ʵ�顢����ʵ�飬�ǵó���ȷʵ����۵�ǰ�ᣬ���Ҫѧ�����ʵ�顢����ʵ�顢����ʵ�飬Ϊѧ�û�ѧ֪ʶ�춨������

| A�� | �ɱ����� | B�� | �ƾ����� | C�� | ��̥��ը | D�� | ����ȼ�� |
| ʵ��װ������ | ���� |
 ���巢��װ�� | ѡ�����巢��װ�õ�������ʲô�� |
 �����ռ�װ�� | ѡ��������ռ�װ�õ�������ʲô�� |
 ����ʵ��װ�� | ��ͼʵ�飬���Թ����д������ݲ����������Թ���û�й۲쵽����ʯ��ˮ����ǣ����ʺ��� |
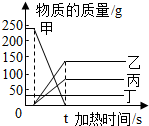 ��ͼΪij��ѧ��Ӧ�и�����������ʱ��Ĺ�ϵͼ�������жϲ���ȷ���ǣ�������
��ͼΪij��ѧ��Ӧ�и�����������ʱ��Ĺ�ϵͼ�������жϲ���ȷ���ǣ�������| A�� | �������Ǵ��� | |
| B�� | �Һͱ��������� | |
| C�� | �÷�Ӧ���ڷֽⷴӦ | |
| D�� | tsʱ��������һ�������Һͱ�������֮�� |
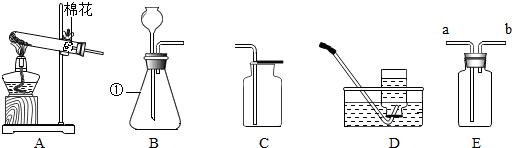


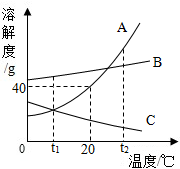 A��B��C�������ʵ��ܽ��������ͼ��ʾ��
A��B��C�������ʵ��ܽ��������ͼ��ʾ��