��Ŀ����
�������г��л�ѧ���õ���ȡ���岿��װ�á���������¸��⣺
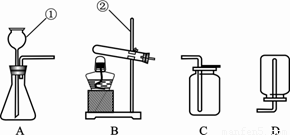
��1��д����ͼ�١����������������ƣ��� __________ ��__________��
��2���Ը������Ϊԭ����ȡ������Ӧѡ��________��________װ�ã�����ĸ������ȡ���ռ�����Ӧ�Ļ�ѧ����ʽ��__________________________________________��
��3������ij��ѧС����ʦ����ѧ����������ʵ��ʱ�����֡�һͬѧ��������װ�������ļ���ƿ��Ѹ�ٷ����ƿ��ʣ�����������ʹ�����ǵ�ľ����ȼ�����Դˣ�С����ʦ�ø�ͬѧ������ƿװ���ƿˮ��������ˮ���ռ�1/2����ƿ������������ƿ�е�����Ҳ��ʹ������ľ����ȼ��
�������з����ռ���1/2����ƿ�����������������������________������ĸ����
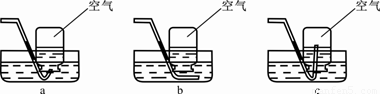
������ˮ���ռ�����ʱ������жϼ���ƿ���������ռ�����
��___________________________________________________________��
�ۿ������������������ԼΪ21%����ʵ���У�����ƿ�ڵ�����Լռ�������______%�������á�ʹ������ľ����ȼ�������鼯��ƿ�г��������ķ������ɿ���
��1���ٳ���©�� ������̨������2��B��C 2KMnO4 K2MnO4+MnO2+O2�� ��3���� a ��
�ڹ۲쵽����ƿ���������������ݳ����� ��60.5
K2MnO4+MnO2+O2�� ��3���� a ��
�ڹ۲쵽����ƿ���������������ݳ����� ��60.5
�����������л�ѧ����2������ķ���װ�ã��ֱ�Ϊ�̹̼�������ȡ���塢��Һ����Ҫ��������ȡ���壬ѡ��������Ƿ�Ӧ�����ͷ�Ӧ���״̬����ʵ���У��ռ�������һ���������ռ����ƿ�������50%������һ���ǿ�����ռ����ƿ�������50%*21%=10.5%���ʼ���ƿ�ڵ�����Լռ�������60.5%

 ��ɢ˼ά�¿���ϵ�д�
��ɢ˼ά�¿���ϵ�д�




 ��1���������������������
��1��������������������� Na2CO3+ CO2�� + H2O
Na2CO3+ CO2�� + H2O
 Na2CO3+CO2��+H2O
Na2CO3+CO2��+H2O