��Ŀ����
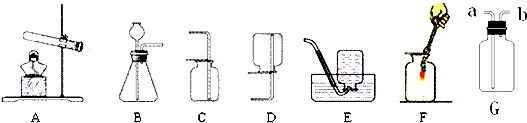
a��b��c��d�dz��л�ѧ�г�����4��Ԫ�أ��й���Ϣ���±���ʾ��A��B��C��D��E��F��G��������4��Ԫ���е�һ�ֻ�������ɵĴ����
��1��A�Ļ�ѧʽ ��
��2�����嵥��D����ȫȼ�պͲ���ȫȼ��ʱ�����ɲ�ͬ������E��F���ù�����ȫȼ������E�Ļ�ѧ����ʽΪ ��
��3����������G���뵽������ˮ�У�����ɫ��������Һ�������Һ��ͨ������E���۲쵽��Һ���
�ǣ��йط�Ӧ�Ļ�ѧ����ʽΪ �� ��ͨ������E����Һ�� �������ֺţ���
������������С ���ܼ��������� ����Һ������С ����������������С��
| Ԫ�� | a | b | c | d |
| �й� ��Ϣ |
�䵥��A���ܶ���С������ | �ؿ��к�������Ԫ�� | �γɻ�������������Ԫ�� | ��Ԫ�ش����������˵Ĺ����������� |
��2�����嵥��D����ȫȼ�պͲ���ȫȼ��ʱ�����ɲ�ͬ������E��F���ù�����ȫȼ������E�Ļ�ѧ����ʽΪ
��3����������G���뵽������ˮ�У�����ɫ��������Һ�������Һ��ͨ������E���۲쵽��Һ���
�ǣ��йط�Ӧ�Ļ�ѧ����ʽΪ
������������С ���ܼ��������� ����Һ������С ����������������С��
���㣺���ʵļ����ƶ�,��ѧʽ����д������,��д��ѧ����ʽ�����ֱ���ʽ�����뷽��ʽ
ר�⣺Ԫ���ƶ���
���������ݵ���A���ܶ���С�����壬��A������������a����Ԫ�أ�b�ǵؿ��к�������Ԫ�أ�����b����Ԫ�أ�c���γɻ�������������Ԫ�أ�����c��̼Ԫ�أ�d�����������˵Ĺ����������У�����d�Ǹ�Ԫ�أ������Ƴ���Ԫ�ؽ�����е����������ͷ�Ӧ��ϵ�Ƶ�ʣ������ʣ�����Ƴ��ĸ������ʴ������е�ת����ϵ��֤���ɣ�
����⣺��1������A���ܶ���С�����壬����A����������ѧʽΪ��H2��
��2�����嵥��D����ȫȼ�պͲ���ȫȼ��ʱ�����ɲ�ͬ������E��F�����֪D��̼��̼����ȫȼ�ջ����ɶ�����̼������ȫȼ��ʱ����һ����̼����ѧ����ʽΪ��C+O2
CO2��
��3������E�ܹ��Ͷ�����̼��Ӧ����̼��Ƴ���������E�������������ƣ���G����Ϊ�����ƣ���������ˮ��Ӧ�������������ƣ�������̼�����������Ʒ�Ӧ����̼��ƺ�ˮ���Ƴ��ĸ������ʾ��������е�ת����ϵ���Ƶ�������������صĻ�ѧ����ʽΪ��CaO+H2O�TCa��OH��2��CO2+Ca��OH��2�TCaCO3��+H2O��
����ʯ��ˮͨ�������̼�������������ƶ�����̼��Ƴ�����ˮ������Һ�����ʵ�����������������������С��ˮ�����ӣ����������غ㶨�ɣ�����������Һ��˵�������˶�����̼���������������ɵ�̼��Ƴ�����������Һ�����������Զ�������Һ��˵��Һ��������Ҳ����٣�����Ӧѡ���٢ڢۢܣ�
�ʴ�Ϊ����1��H2��
��2��C+O2
CO2��
��3��CaO+H2O�TCa��OH��2��CO2+Ca��OH��2�TCaCO3��+H2O���٢ڢۢܣ�
��2�����嵥��D����ȫȼ�պͲ���ȫȼ��ʱ�����ɲ�ͬ������E��F�����֪D��̼��̼����ȫȼ�ջ����ɶ�����̼������ȫȼ��ʱ����һ����̼����ѧ����ʽΪ��C+O2
| ||
��3������E�ܹ��Ͷ�����̼��Ӧ����̼��Ƴ���������E�������������ƣ���G����Ϊ�����ƣ���������ˮ��Ӧ�������������ƣ�������̼�����������Ʒ�Ӧ����̼��ƺ�ˮ���Ƴ��ĸ������ʾ��������е�ת����ϵ���Ƶ�������������صĻ�ѧ����ʽΪ��CaO+H2O�TCa��OH��2��CO2+Ca��OH��2�TCaCO3��+H2O��
����ʯ��ˮͨ�������̼�������������ƶ�����̼��Ƴ�����ˮ������Һ�����ʵ�����������������������С��ˮ�����ӣ����������غ㶨�ɣ�����������Һ��˵�������˶�����̼���������������ɵ�̼��Ƴ�����������Һ�����������Զ�������Һ��˵��Һ��������Ҳ����٣�����Ӧѡ���٢ڢۢܣ�
�ʴ�Ϊ����1��H2��
��2��C+O2
| ||
��3��CaO+H2O�TCa��OH��2��CO2+Ca��OH��2�TCaCO3��+H2O���٢ڢۢܣ�
���������⿼���˳���Ԫ���Լ����ʵ��ƶϣ���ɴ��⣬�����������е�֪ʶ���У�Ҫ��ͬѧ��������ʵ����ʣ��Լ�����֮���ת�����Ա����Ӧ�ã�

��ϰ��ϵ�д�
�����Ŀ
���ҷ�Ӧ���Ʊ�ȼ�ϱ�������ʾ��ͼ���£�����˵��������ǣ�������
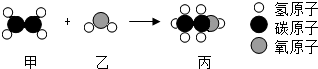

| A���÷�Ӧ�з��Ӻ�ԭ�ӵ���������˸ı� |
| B���÷�Ӧ���ڻ��Ϸ�Ӧ |
| C���ס������л������������ |
| D���ס����ֱ�����������ȫȼ�գ�������ͬ |
 ��1��ijͬѧ����ͼװ�ö������غ㶨�ɽ���ʵ��̽������Ӧǰ��ƽƽ�⣬Ȼ�����ᵹ���ձ�����̼���Ƴ�ַ�Ӧ���ٳ������۲쵽��Ӧ����ƽ��ƽ�⣬��ƽָ����
��1��ijͬѧ����ͼװ�ö������غ㶨�ɽ���ʵ��̽������Ӧǰ��ƽƽ�⣬Ȼ�����ᵹ���ձ�����̼���Ƴ�ַ�Ӧ���ٳ������۲쵽��Ӧ����ƽ��ƽ�⣬��ƽָ����
 ��ѧ������ϢϢ��أ�
��ѧ������ϢϢ��أ�