��Ŀ����
��ͼ��ʾ��þԪ����Ԫ�����ڱ��е���Ϣ��ԭ�ӵ�ԭ�ӽṹʾ��ͼ���ݴ��жϣ�����˵������ȷ����
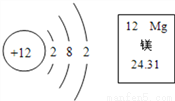
A. þԭ�ӵ����ԭ������Ϊ24.31
B. �ڻ�ѧ��Ӧ�У�þԭ�������γ�������
C. ��Ԫ���γɵ���������MgNO3
D. þԭ�Ӻ�����l 2������
C ��������A������Ԫ�����ڱ��е�һ���л�ȡ����Ϣ����֪Ԫ�ص����ԭ������Ϊ24.31����ȷ��B��þԭ�ӵ�������������2���ڻ�ѧ��Ӧ����ʧȥ2�����Ӷ��γ������ӣ���ȷ��C����Ԫ���γɵ���������Mg��NO3��2������D������Ԫ�����ڱ��е�һ���л�ȡ����Ϣ����Ԫ�ص�ԭ������Ϊ12������ԭ������=�����������Ԫ�ص�ԭ�ӵ�������Ϊ12������þԭ�ӵĽṹʾ��ͼ����������Ϊ12����ȷ����ѡC��
��ϰ��ϵ�д�
�����Ŀ
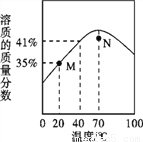
Ϊ��ȥ���������е����ʣ�������Ϊ���ʣ������в����������ܴﵽĿ�ĵ��ǣ� ��
ѡ �� | ���ʣ����ʣ� | ������ʽ |
A | Cu(Fe) | ������ϡ���ᣬ��ַ�Ӧ����� |
B | CO(O2) | ����ͨ������ͭ�� |
C | NaCl��Һ��Na2CO3�� | ��������ǡ�ò��ٲ�������Ϊֹ |
D | FeSO4��Һ��CuSO4�� | ����������м����ַ�Ӧ����� |



 K2MnO4+MnO2+O2��
K2MnO4+MnO2+O2��