��Ŀ����
�����ӽ���ʶ�Ϳ�������������ѧϰ��ѧ����Ҫ˼ά������
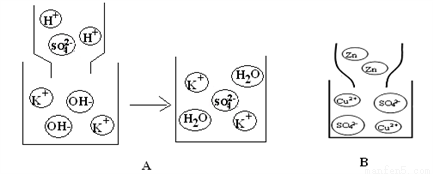
��1����ͼ��ij��Ӧ����ʾ��ͼ��

B���е���������___________������ţ��ٻ�����ڵ��ʡ��۴�����ܻ������˷�Ӧ�Ļ�ѧ����ʽΪ_____________________________����ͼ˵����ѧ��Ӧǰ�����ı������_________������ӡ�����ԭ�ӡ������ӡ�����
��2��д����ͼA��ʾ�ķ�Ӧ��ѧ����ʽ ________________________________ ���÷�Ӧǰ����Һ���ٵ������� _____________����ͼB�з�Ӧ��Һ��Ϊ��ɫʱ����ʱ��Һ�д��ڵ�������________________��
��ϰ��ϵ�д�
 ϰ�⾫ѡϵ�д�
ϰ�⾫ѡϵ�д�
�����Ŀ








