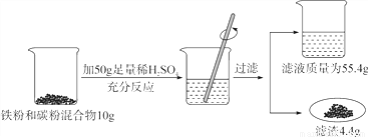
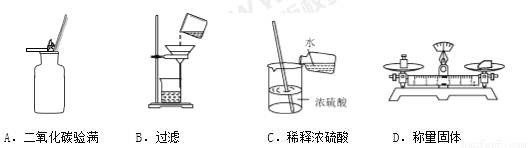
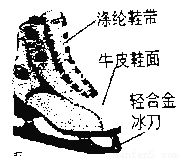
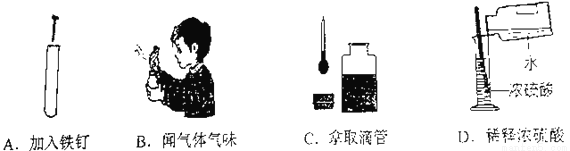
��Ŀ����
��������ʱ��Ҫ������������������(CaO2)��һ�ֳ������������ȡ�����������ˮ��Ӧ����ʽΪ��2CaO2+2H2O=2Ca(OH)2+O2
(1)��Ʒ��CaO2������
(2)�����Ͽ����Ƶ�����Ϊ���ٵ�����?
144g��32g ����������1��150g��Ʒ�й������Ƶ�����Ϊ150g=144g�� ��2���裺�����Ͽ����Ƶ�����������Ϊx 2CaO2+2H2O=2Ca(OH)2+O2 144 32 144g x =���x=32g ����Ʒ��CaO2������Ϊ144g������������������Ϊ32g��
��ϰ��ϵ�д�
 ʱ�����������ҵԭ���ܳ�����ϵ�д�
ʱ�����������ҵԭ���ܳ�����ϵ�д� ����νӽ̲���ĩ���Ԥϰ�人������ϵ�д�
����νӽ̲���ĩ���Ԥϰ�人������ϵ�д� ������ҵ��ٳɳ����½������������ϵ�д�
������ҵ��ٳɳ����½������������ϵ�д�
�����Ŀ