��Ŀ����
A��B��C��D��E�������ʾ�Ϊ���л�ѧ�г����Ļ��������֮��Ĺ�ϵ����ͼ��ʾ���֮�����߱�ʾ�����ܷ�����ѧ��Ӧ��A��C��E��Ϊ�����A�������������Ҫ���壻��ʵ������B��������ȡA����D������������A��C��E��Ӧ������D��
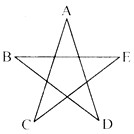
(1)A�Ļ�ѧʽ��____��D�Ļ�ѧʽ��____��
(2)B������ȡA֮�����һ����;��______________________��
(3)C��E��Ӧ�Ļ�ѧ����ʽΪ________________________���÷�Ӧ�Ļ���������______________��
(2)B������ȡA֮�����һ����;��______________________��
(3)C��E��Ӧ�Ļ�ѧ����ʽΪ________________________���÷�Ӧ�Ļ���������______________��
(1)CO2��Ca(OH)2
(2)���ڽ���������⣨������ҩ��ɰ��������ȣ�
(3)CaO+H2O==Ca(OH)2�����Ϸ�Ӧ
(2)���ڽ���������⣨������ҩ��ɰ��������ȣ�
(3)CaO+H2O==Ca(OH)2�����Ϸ�Ӧ

��ϰ��ϵ�д�
 ��һ����ͬ���ɽ�����ϵ�д�
��һ����ͬ���ɽ�����ϵ�д� ������Ӧ���ϵ�д�
������Ӧ���ϵ�д� ��ʦ�㾦�ִʾ��ƪϵ�д�
��ʦ�㾦�ִʾ��ƪϵ�д�
�����Ŀ
 ��֪��A����Է���������С�������D������л�������A��B��C��D��E��������֮��ı仯��ϵ���ش��й����⣮
��֪��A����Է���������С�������D������л�������A��B��C��D��E��������֮��ı仯��ϵ���ش��й����⣮
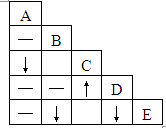 ��A��B��C��D��E��ƿʧȥ��ǩ����Һ����ƿ������Na2CO3��BaCl2��Ca��NO3��2��ϡ���ᡢAgNO3��Һ�е�һ�֣��ֽ�ƿ����Һ��ȡ���������в���������ϵ�ʵ�飬ʵ��������ͼ��˵�������С���ʾ���������ɣ�����ʾ�г������ɣ�-��ʾ��������������ܻ��������ɣ��ո��ʾʵ��û����
��A��B��C��D��E��ƿʧȥ��ǩ����Һ����ƿ������Na2CO3��BaCl2��Ca��NO3��2��ϡ���ᡢAgNO3��Һ�е�һ�֣��ֽ�ƿ����Һ��ȡ���������в���������ϵ�ʵ�飬ʵ��������ͼ��˵�������С���ʾ���������ɣ�����ʾ�г������ɣ�-��ʾ��������������ܻ��������ɣ��ո��ʾʵ��û����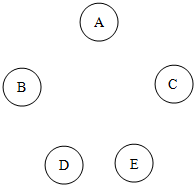 ��2013?�ൺ������A��B��C��D��E�������ʣ��ֱ�Ϊ���ᡢ����ͭ���������ơ����ۺ��������е�һ�֣����У�A��Ũ��Һ����ɫҺ�壬�д̼�����ζ���ڿ����л��γɰ�����BΪ���ʣ�D�ǡ�������Һ������Ҫ�ɷ�֮һ��E����ʯ�ҵ���Ҫ�ɷ֣���ش��������⣺
��2013?�ൺ������A��B��C��D��E�������ʣ��ֱ�Ϊ���ᡢ����ͭ���������ơ����ۺ��������е�һ�֣����У�A��Ũ��Һ����ɫҺ�壬�д̼�����ζ���ڿ����л��γɰ�����BΪ���ʣ�D�ǡ�������Һ������Ҫ�ɷ�֮һ��E����ʯ�ҵ���Ҫ�ɷ֣���ش��������⣺