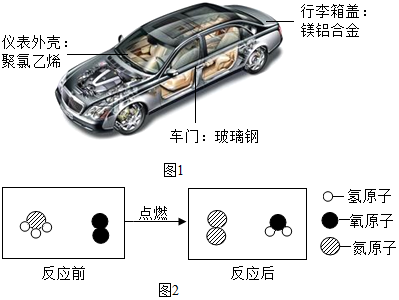
��Ŀ����
9���������кͷ�Ӧ����ʵ��������Һ�д��������ƶ���H+�ͼ���Һ�д��������ƶ���OH-�������ˮ���ӵĹ��̣����������ϡ������ϡ����������Һ��Ӧ��ʵ�飬�ش����⣮��ʵ��һ���÷�̪��Һ
��1����һ�Թ��е���Լ2mLϡ����������Һ���μ�3�η�̪��Һ��������ε���ϡ���ᣬ�ߵα���ֱ����Һ�պñ�Ϊ��ɫΪֹ��˵������֮�䷢�����кͷ�Ӧ��
��ʵ��������¶ȼ�
��2����һ�ձ��м���һ������ϡ�������Ƽ�Һ����������ټ���ϡ���ᣬ�������ֻ������Է�Ӧ��������Һ���¶�ʵʱ�ⶨ���õ���������ͼ1��
��Լ0-50s�¶���������Ϊ�кͷ�Ӧ���ȣ�50s����Һ�¶��½���ԭ���Ƿ�Ӧ����������ɢʧ��������ϡ��������ȴ���ã�
�ڲ������������ƹ�����ϡ���ᷴӦ�ⶨ�кͷ�Ӧ���ȵ�ԭ�����������ƹ����ܽ���ˮʱ����ȣ�

��ʵ��������pH��
��3����һ��ƿ�м���20g��������Ϊ4.00%��ϡ����������Һ����������ټ���һ������������ϡ���ᣬ�������ֻ������Է�Ӧ��������Һ��pH�ⶨ���õ���������ͼ2����ʽ���㣺
����ȡϡ�������������Ϊ���٣�����ȷ��0.0l%����ͬ��
�ڵ�ǡ����ȫ��Ӧʱ������Һ��������������Ϊ���٣�
���� ��1������ָʾ���ı�ɫ���������
��2���������ڷ�Ӧ�Ѿ���ֹ��������ɢ�������������ƹ�������ˮ����ȣ�
��3���۲�ͼʾ�������μӵ�ϡ�������������Һ��pH�仯�ĺ�����ϵͼ���ڼ���20gϡ����������ҺpH=7ʱ����������������ǡ�÷�Ӧ�����ݷ�Ӧ�Ļ�ѧ����ʽ���������������Ƶ�������������ϡ�������Ȼ�������������ɵ��Ȼ��Ƶ������������������������㹫ʽ���ϡ�������ʵ�����������������Һ���Ȼ��Ƶ�����������
��� �⣺��1����̪�����죬�����Ժ����Ի�����Ϊ��ɫ����������������Һ�е��뼸����ɫ��̪��������Һ�ʺ�ɫ������ε���ϡ���ᣬ�ߵα�����ǡ����ȫ��Ӧʱ����Һ����ɫ��
��2����50s��Ӧ����������ɢʧ���Լ�������ϡ��������ȴ���ã���Һ�¶��½���
�������������ƹ����ܽ���ˮʱ����ȣ����ж��кͷ�Ӧ�Ƿ���ȣ�
��3�����Ȼ��������Ϊx�������Ȼ��Ƶ�����Ϊy������
NaOH+HCl=NaCl+H2O
40 36.5 58.5
20g��4.00% x y
$\frac{40}{20g��4.00%}=\frac{36.5}{x}=\frac{58.5}{y}$
x=0.73g��y=1.17g
����ȡϡ�������������Ϊ$\frac{0.73g}{20g}��100%$=3.65%��
�ڵ�ǡ����ȫ��Ӧʱ������Һ��������������Ϊ$\frac{1.17g}{20g+20g}��100%$=2.93%��
�ʴ�Ϊ����1���ޣ�
��2���ٷ�Ӧ����������ɢʧ��������ϡ��������ȴ���ã�
���������ƹ����ܽ���ˮʱ����ȣ�
��3����3.65%�� ��2.93%��
���� ������һ���кͿ����кͷ�Ӧ����Ŀ�����ͼ��ļ����⣬����ʱ�ҵ�pH=7��ǡ�÷�Ӧ�㣬�����û�ѧ����ʽ�ļ�����н���ǽ����ͻ�ƿڣ�

| A�� | ά����C�������� | B�� | ά����C��̼Ԫ�ص������������ | ||
| C�� | һ��ά����C��������20��ԭ�� | D�� | ά����C��Ԫ��������Ϊ��3��4��3 |
| A�� | ��һ��̼���� | B�� | ����̼Ԫ�غ���Ԫ�� | ||
| C�� | ���е����� | D�� | ���п�ȼ�� |
| ���� | X | Y | M | N |
| ��Ӧǰ��������/g | 1.7 | 2.2 | 7.9 | 0.9 |
| ��Ӧ����������/g | m | 6.6 | 0 | 2.7 |
| A�� | m ��ֵΪ1.7 | |
| B�� | ����X�������� | |
| C�� | �μӻ�ѧ�仯������Y��N��������Ϊ22��9 | |
| D�� | �÷�Ӧһ���Ǹ�����طֽ������� |
| A�� | ʳ��ˮ��ϡ���pH��ֽ | |
| B�� | �������ƹ���������粒��壺ˮ | |
| C�� | ������̼�Ͷ��������������Һ | |
| D�� | ̼������Һ������������Һ����̪��Һ |
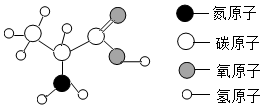
| A�� | ��������һ���л��߷��ӻ����� | |
| B�� | ��������̼����Ԫ�ظ�����Ϊ3��1 | |
| C�� | ����������Ԫ�ص�����������С | |
| D�� | ��������15��̼ԭ�ӡ�22����ԭ�Ӻ�5����ԭ�ӹ��� |
| A�� | ��ʯ�����ڸ����������� | B�� | ����������ȼ�� | ||
| C�� | ͭ˿�������� | D�� | ������̼������������� |
 �����г�����Ӫ�����������ǵij��У��ڸ����г��Ͽ��ṩ����ʳƷ������ˮ����֭��ţ�̡����ɡ�ˮ���ȣ�
�����г�����Ӫ�����������ǵij��У��ڸ����г��Ͽ��ṩ����ʳƷ������ˮ����֭��ţ�̡����ɡ�ˮ���ȣ�