��Ŀ����
С����ʵ���ҷ���һƿδ֪Ũ�ȵ�����������Һ��Ϊ�����Ժ�ʹ�ã�������Ũ�Ƚ����˲ⶨ��ȡ20.0 g������������Һ���ձ��У���εμ�������������Ϊ7.3%��ϡ���ᣬ����ʱ�Է�Ӧ�����Һ��pH��(һ�ֲⶨ��ҺpH������)�ⶨ��Һ��pH�������������±���
| ����ϡ���� | |||||
| ������ | 9.6 | 9.8 | 9.9 | 10.0 | 10.1 |
| ��Һ��pH | 12.4 | 12.1 | 11.8 | 7.0 | 2.2 |
�Իش�
(1)���μ�ϡ���������Ϊ9.8 gʱ����Һ�е�������__ __��__��
(2)������������������Һ�����ʵ�����������
_
�⣺��20.0 g����������Һ�����ʵ�����Ϊx
HCl �� NaOH===NaCl �� H2O
36��5����������40
10 g��7.3% x
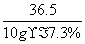 ��
��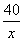
x��0.8 g
����������Һ�����ʵ�����������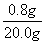 ��100%�� 4%
��100%�� 4%

��ϰ��ϵ�д�
�����Ŀ
 /g
/g �ʵ����������������ʵ���(����)��
�ʵ����������������ʵ���(����)��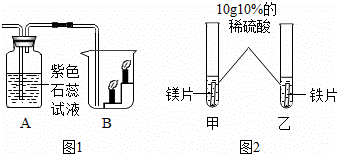
 �����л�ѧ��Ӧ��2NaN3 �� NH4NO3 === 3X���� 2Na �� N2O���� 2H2O����X�Ļ�ѧʽΪ( )
�����л�ѧ��Ӧ��2NaN3 �� NH4NO3 === 3X���� 2Na �� N2O���� 2H2O����X�Ļ�ѧʽΪ( ) ��
��