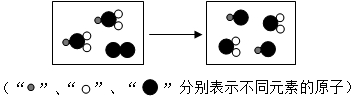
��Ŀ����
|
 |
|
|
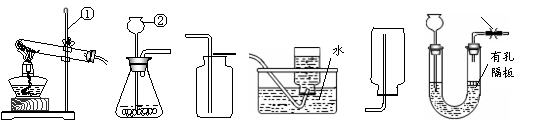
��1����Ţ٢ڵ��������ƣ��� ���� ��
��2��ʵ���� ѡ��A��Dװ����ȡO2�������ռ����ʱ��Ӧ�� �����ˮ
ѡ��A��Dװ����ȡO2�������ռ����ʱ��Ӧ�� �����ˮ ��ȡ�������ܡ���Ϩ��ƾ��ơ�����
��ȡ�������ܡ���Ϩ��ƾ��ơ�����
��3��ʵ���ҿ���(NH4)2SO4�������ʯ�һ�ϼ�����ȡNH3��Ӧѡ����װ��Ϊ ��ѡ����ţ���
 ��4��ѡ����װ��B����
��4��ѡ����װ��B���� ��װ�� ��ѡ����ţ���������ʵ������ȡCO2����������װ����B��ΪF�����ŵ���
��װ�� ��ѡ����ţ���������ʵ������ȡCO2����������װ����B��ΪF�����ŵ���  ��
��
��Ϊ�˵õ������������CO2������װ�ã�����ͼ��
�ĵ��ܰ�������������˳���� ��ѡ����ĸ����
 a��1��2��4��3 b��2��1��3��4
a��1��2��4��3 b��2��1��3��4
c��4��3��1��2 d��3��4��2��1
�û�ѧ��Ӧ����ʽ��ʾ����NaHCO3��Һ���� �� ��
�� ��
��д���ó���ʯ��ˮ����CO2�Ļ�ѧ��Ӧ����ʽ ��
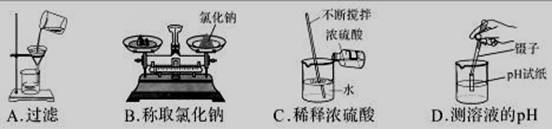
��1��������̨ �ڳ���©�� ��2����ˮ��ȡ�������ܡ�
��3��A��4�� C ���Ʒ�Ӧ�ķ�����ֹͣ����˼����������𰸾����֣���
C ���Ʒ�Ӧ�ķ�����ֹͣ����˼����������𰸾����֣���
�� c NaHCO3 + HCl = NaCl + H2O + CO2��
�� CO2 + Ca(OH)2 = CaCO3��+ H2O

ά����C��������Ѫ�ᣬ������ˮ�������ԣ��ױ�����������ⷴӦ������ȱ��ά����C�����������ּ�����ij�о���ѧϰС���ά����C���о����£�
[ʵ��Ŀ��]��֤ά����C�������ԡ�
[ʵ�鷽��]�ṩ�Լ���ά����CƬ������ˮ����ɫʯ����Һ����ɫ��̪��Һ������������Һ��pH��ֽ����ʵ��������ѡ��
���������Լ��������������������ַ�������д�����ܹ۲쵽��ʵ������
| ʵ�鷽�� | ʵ������ |
| ������1�� | |
| ������2�� |
 ClO��ClԪ��
ClO��ClԪ�� �Ļ��ϼ���_____________��
�Ļ��ϼ���_____________�� ����ɱ������̣�������Ϊ________________________________________________________________________��
����ɱ������̣�������Ϊ________________________________________________________________________�� ���·�Ӧ��Ca(ClO)2+X+H2O��CaCO3��+2HClO,��X�Ļ�ѧʽΪ__________��
���·�Ӧ��Ca(ClO)2+X+H2O��CaCO3��+2HClO,��X�Ļ�ѧʽΪ__________��