��Ŀ����
��������װ��ͼ1���ش��й����⣺
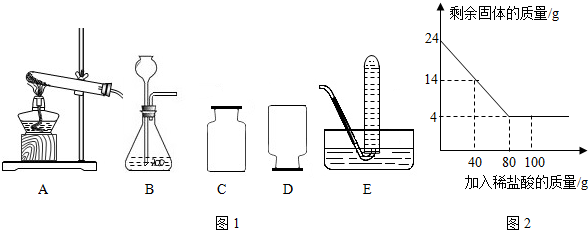
��1���ø��������ȡ�����Ļ�ѧ����ʽΪ ����ѡ��A���÷�Ӧ�ķ���װ�ã���װ��A�е��Թܿڻ�Ҫ
��һ�� �����Թܿ���������б��ԭ���� ��
��2��ʵ������ȡij����ʱ��������DҲ����Eװ���ռ����ɴ��Ʋ�����������е����������� ������ţ���
a����������ܶȴ��ڿ��� b��������������ˮ c����������ܶ�С�ڿ���
��3��ʵ������ȡCO2��������Ҫ���裺�����Ӻ�װ�ã��� �������Թ���С�ķ��뼸С��ʯ��ʯ�������Թ���С��ע������ϡ������ռ����������壻����ȼ�յ�ľ�������ռ�ƿ�Ƿ��ռ���CO2��
��4����Ҫ��װ��E�ռ�CO2���壬Ҫ��ˮ������ˮ���������� ��
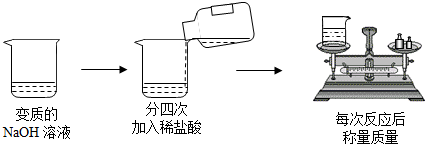
��5��ij��ѧ��ȤС��Ϊ�˲ⶨ���ص�ʯ��ʯ��CaCO3������������������������ʵ�飺ȡ24g��Ʒ�����ձ��У�����������100��һ������������ϡ���ᣨ���ʲ�����ˮҲ�����ᷴӦ�����������������������ʣ������������ϵ��ͼ2��ʾ�������ͼ�е����ݽ��м��㣺
��a��ʯ��ʯ��Ʒ��CaCO3������Ϊ �ˣ�
��b��24��ʯ��ʯ��Ʒ�������ַ�Ӧ�����������ٿ˶�����̼��������ݻ�ѧ����ʽд�������ļ��㲽�裩

��1���ø��������ȡ�����Ļ�ѧ����ʽΪ
��һ��
��2��ʵ������ȡij����ʱ��������DҲ����Eװ���ռ����ɴ��Ʋ�����������е�����������
a����������ܶȴ��ڿ��� b��������������ˮ c����������ܶ�С�ڿ���
��3��ʵ������ȡCO2��������Ҫ���裺�����Ӻ�װ�ã���
��4����Ҫ��װ��E�ռ�CO2���壬Ҫ��ˮ������ˮ����������
��5��ij��ѧ��ȤС��Ϊ�˲ⶨ���ص�ʯ��ʯ��CaCO3������������������������ʵ�飺ȡ24g��Ʒ�����ձ��У�����������100��һ������������ϡ���ᣨ���ʲ�����ˮҲ�����ᷴӦ�����������������������ʣ������������ϵ��ͼ2��ʾ�������ͼ�е����ݽ��м��㣺
��a��ʯ��ʯ��Ʒ��CaCO3������Ϊ
��b��24��ʯ��ʯ��Ʒ�������ַ�Ӧ�����������ٿ˶�����̼��������ݻ�ѧ����ʽд�������ļ��㲽�裩
���㣺��������ķ���װ�ú��ռ�װ����ѡȡ����,ʵ������ȡ�����ķ�Ӧԭ��,��ȡ������̼�IJ��������ע���,���ݻ�ѧ��Ӧ����ʽ�ļ���
ר�⣺�йػ�ѧ����ʽ�ļ���,���������ʵ�����Ʒ������顢�����뾻��
��������ȡװ�ð������ȺͲ���������֣������˫��ˮ�Ͷ��������������Ͳ���Ҫ���ȣ�����ø�����ػ����������������Ҫ���ȣ����ȸ������ʱ���Թܿ�Ҫ��һ��������Ϊ�˷�ֹ������ط�ĩ���뵼�ܣ��������ܶȱȿ������ܶȴ�������ˮ��������������ſ���������ˮ���ռ���ʵ������ȡCO2�����ڳ����£���̼��ƺ����ụ�ཻ���ɷ������Ȼ��ƺ�ˮ�Ͷ�����̼����˲���Ҫ���ȣ�������̼������ˮ���ܶȱȿ������ܶȴ����ֻ���������ſ������ռ���ʵ������ȡij����ʱ��������DҲ����Eװ���ռ����ɴ��Ʋ�����������е����������ǣ�������������ˮ����������ܶ�С�ڿ�����ʵ������ȡCO2����Ҫ��Ҫ�����ǣ����Ӻ�װ�ã����װ�õ������ԣ���Ҫ��װ��E�ռ�CO2���壬Ҫ��ˮ������ˮ���������ǣ�������ܽ�����¶ȵ����߶���С������CO2��������ˮ�е��ܽ�Ƚ�С����ͼ���֪��ʯ��ʯ��Ʒ��CaCO3������=24g-4g=20g��Ȼ����ݻ�ѧʽ�ļ����������̼��������
����⣺��1������ø�����ػ����������������Ҫ���ȣ����ȸ������ʱ���Թܿ�Ҫ��һ��������Ϊ�˷�ֹ������ط�ĩ���뵼�ܣ��Թܿ���������б��ԭ���ǣ���ֹ����ˮ����ʹ�Թ�ը�ѣ��ʴ�Ϊ��2KMnO4
K2MnO4+MnO2+O2�� �� ��ֹ����ˮ����ʹ�Թ�ը��
��2��ʵ������ȡij����ʱ��������DҲ����Eװ���ռ����ɴ��Ʋ�����������е����������ǣ�������������ˮ����������ܶ�С�ڿ������ʴ�Ϊ��bc
��3��ʵ������ȡCO2����Ҫ��Ҫ�����ǣ����Ӻ�װ�ã����װ�õ������ԣ��ʴ�Ϊ�����װ�õ�������
��4����Ҫ��װ��E�ռ�CO2���壬Ҫ��ˮ������ˮ���������ǣ�������ܽ�����¶ȵ����߶���С������CO2��������ˮ�е��ܽ�Ƚ�С���ʴ�Ϊ��������ܽ�����¶ȵ����߶���С������CO2��������ˮ�е��ܽ�Ƚ�С������CO2��ˮ�е��ܽ�ȣ�
��5����ͼ���֪��ʯ��ʯ��Ʒ��CaCO3������=24g-4g=20g���ʴ�Ϊ����a��20
��b���⣺�����CO2������Ϊx
CaCO3+2HCl=CaCl2+H2O+CO2��
100 44
20g x
=
x=8.8g
�𣺲���CO2������Ϊ8.8g
�ʴ�Ϊ����b��8.8g��
| ||
��2��ʵ������ȡij����ʱ��������DҲ����Eװ���ռ����ɴ��Ʋ�����������е����������ǣ�������������ˮ����������ܶ�С�ڿ������ʴ�Ϊ��bc
��3��ʵ������ȡCO2����Ҫ��Ҫ�����ǣ����Ӻ�װ�ã����װ�õ������ԣ��ʴ�Ϊ�����װ�õ�������
��4����Ҫ��װ��E�ռ�CO2���壬Ҫ��ˮ������ˮ���������ǣ�������ܽ�����¶ȵ����߶���С������CO2��������ˮ�е��ܽ�Ƚ�С���ʴ�Ϊ��������ܽ�����¶ȵ����߶���С������CO2��������ˮ�е��ܽ�Ƚ�С������CO2��ˮ�е��ܽ�ȣ�
��5����ͼ���֪��ʯ��ʯ��Ʒ��CaCO3������=24g-4g=20g���ʴ�Ϊ����a��20
��b���⣺�����CO2������Ϊx
CaCO3+2HCl=CaCl2+H2O+CO2��
100 44
20g x
| 100 |
| 44 |
| 20g |
| x |
�𣺲���CO2������Ϊ8.8g
�ʴ�Ϊ����b��8.8g��
��������������Ҫ�������������ȡװ�ú��ռ�װ�õ�ѡ��ͬʱҲ�����˻�ѧ����ʽ����д��ע��������йصļ���ȣ��ۺ��ԱȽ�ǿ���������ȡװ�õ�ѡ���뷴Ӧ���״̬�ͷ�Ӧ�������йأ�������ռ�װ�õ�ѡ����������ܶȺ��ܽ����йأ����������п�����Ҫ����֮һ����Ҫ������ʵ�����У�

��ϰ��ϵ�д�
�����Ŀ
�������ʳ��ڱ�¶�ڿ����У���ѧ�仯ʹ�������ӵ��ǣ�������
| A��Ũ���� | B��Ũ���� |
| C���ռ���� | D����ʯ�� |
���������мȲ������ڼ���Ҳ������������Ӧ�������ǣ�������
| A���Թ� | B�������� |
| C����Ͳ | D����ƿ |
����ֱ���ھƾ����ϼ��ȵIJ��������ǣ�������
| A��ȼ�ճ� | B�������� |
| C���Թ� | D���ձ� |