��Ŀ����
(14��)ˮ��ʵ���Һ��ճ��������кܶ���;��
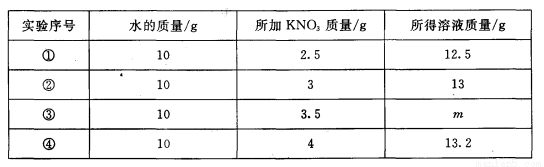
I����Һ���ճ������еij������ʣ�ˮ����õ��ܼ���
20��ʱ�����������ļס������ֹ������ʣ��ֱ���뵽ʢ��100gˮ���ձ��У���ֽ����������ͼ1�����ȵ�50��ʱ������ͼ2���ס����������ʵ��ܽ��������ͼ3������ͼʾ�ش��������⣺
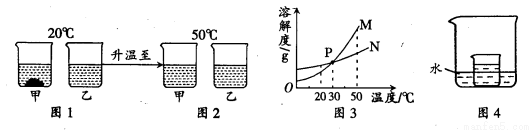
��1��ͼ1��һ��Ϊ������Һ����
��2��ͼ3�б�ʾ�ҵ��ܽ�������� ��P���ʾ�ĺ����� ��
��3����ͼ4��ʾ��ʵ���У�С�ձ���ʢ�ŵ���20��ʱ�����ʵı�����Һ��ijͬѧҪʹС�ձ����й�������ʱ����Ӧ����ձ���ˮ�м������������е� ������ĸ����
A�������� B���Ȼ��� C���������� D�������
��ˮ�ڹ�ҵ������Ҳ����Ҫ��
�Ե��ʳ��ˮΪ������ȡ�����Ȳ�Ʒ�Ĺ�ҵ��Ϊ���ȼҵ��������Ŀǰ��ѧ��ҵ����Ҫ֧��֮һ����ⱥ��ʳ��ˮ���Եõ����ֻ�����Ʒ����Ҫ�������£�
[���Ͽ���ʯ��ˮ�е�������һ�ּ��Ca(OH)2]
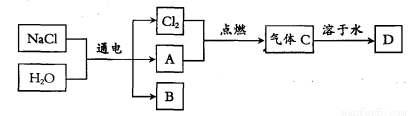
��4����֪����A���ܶ���С�����壬B��һ�ּD��һ���ᡣд������������D��Ӧ��������A�Ļ�ѧ����ʽ�� ��
��5��B�Ļ�ѧʽΪ ��
��6����ҵ�ϵ��ʳ��ˮ������ʽ��Ĥ�����н��У�������Cl2������A�������ݳ����ֱ��ռ������档
����֪20�棬S(NaCl)��36g���������£�68 kg����ʳ��ˮ�����ʵ�����Ϊ ��
�ڵ��һ��ʱ�䣬���ռ�������������Ϊ0.71 kgʱ����ʽ�������ɵ�B������ ��
��0.71 kg������A��ȫ��Ӧ��������C������һ������ˮ���γ���D������D�е�������������Ϊ36.5%��������ˮ������Ϊ ��
��1���� ��2��N 30��ʱM��N���ܽ����� ��3��D ��4��2Al + 6HCl=2AlCl3 + 3H2��
��5����18kg ��0.8kg ��1.27kg
��������
�����������1���ж���Һ�Ƿ͵ķ�������Һ����δ�ܽ�����ʣ���ͼ1��һ��Ϊ������Һ���Ǽ�
��2��������20��ʱ���������ļס������ֹ������ʣ��ֱ���뵽ʢ��100gˮ���ձ��У�������ܽ�ĸ��٣�˵����20��ʱ�����ܽ��ҪС���ҵ��ܽ�ȣ���ͼ3�б�ʾ�ҵ��ܽ��������N�������н���P���ʾ�ĺ����ǣ�30��ʱM��N���ܽ�����
��3�����ڼ��ܽ�����¶ȵ����߶�����Ҫʹ20��ʱ�����ʵı�����Һ�������壬���Խ����¶ȣ����������ˮ����������ʹ�¶Ƚ��ͣ���ѡD
��4����ΪA���ܶ���С�����壬��������������������ȼ����������CΪ�Ȼ��⣬����ˮ��õ����ᣬ�ʽ���������D��Ӧ��������A�Ļ�ѧ����ʽ��2Al + 6HCl=2AlCl3 + 3H2��
��5������֪20�棬S(NaCl)��36g����ʾ��100gˮ�дﵽ����״̬�ܽ���Ȼ�������Ϊ36g���������£�68 kg����ʳ��ˮ�����ʵ�����Ϊ18kg
�ڸ�������ķ�����֪B��NaOH��Ȼ���ٸ��ݻ�ѧ����ʽ��2NaCl+2H2Oͨ��2NaOH+H2��+Cl2����NaOH��Cl2��������ϵ�������NaOH������
��������
��NaOH������Ϊx
2NaCl+2H2Oͨ��2NaOH+H2��+Cl2��
71
X 0.71 kg
80:71=x��0.71 kg
x=0.8kg
�����ȸ��ݻ�ѧ��Ӧ��H2+Cl2��ȼ2HCl��Cl2��HCl��������ϵ�����HCl���������ٸ����������������ļ������ʽ����
��������
��HCl������Ϊx
H2+Cl2��ȼ2HCl
73
0.71kg x
71:73=0.71kg ��x
x=0.73 kg
�����ˮ������Ϊy����ʽΪ��0.73 kg����0.73 kg+y��=36.5%
y=1.27kg
���㣺������Һ����������Һ���жϣ��ܽ�����ߵ����壬���ݻ�ѧ����ʽ���㣬��Һ������

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�