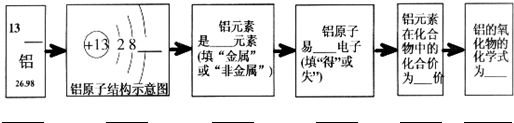
��Ŀ����
ij���������ɲɹ������ռ��в��ֱ��ʣ���������ԱΪ�˲ⶨ�������������ⶨ�ռ�Ĵ��ȣ�������Ʒ��ֻ���������ƺ�̼���������ֳɷ֣���̼�����DZ��ʲ����ģ�����������ʵ�飺ȡ�ռ���Ʒ20g�����Ĵμ��뵽36.5gһ��Ũ�ȵ�ϡ�����У��������±���ʾ
������ṩ�����ݣ��ش��������⣺
��1��������m����ֵΪ ��
��2��������ռ���Ʒ���ռ�Ĵ��ȣ�
| ʵ����� | 1 | 2 | 3 | 4 |
| ÿ�μ�����Ʒ������/g | 5 | 5 | 5 | 5 |
| ��Ӧ��ʣ�����ʵ�������/g | 40.4 | 44.3 | m | 54.3 |
��1��������m����ֵΪ
��2��������ռ���Ʒ���ռ�Ĵ��ȣ�
���㣺���ݻ�ѧ��Ӧ����ʽ�ļ���
ר�⣺�ۺϼ��㣨ͼ���͡������͡��龰�ͼ����⣩
��������1�����ݱ�������ݷ���ÿ�β��������������֪��
��2������������������������Ʒ��̼���Ƶ����������������Ʒ���������Ƶ�����������
��2������������������������Ʒ��̼���Ƶ����������������Ʒ���������Ƶ�����������
����⣺��1���ɱ������ݿ�֪���ڶ��β���CO2��������36.5g+5g+5g-44.3g=2.2g�����Ĵβ���������̼�������ǣ�36.5g+5g+5g+5g+5g-54.3g=2.2g���ʵ����β���������̼������Ҳ��2.2g����m��ֵΪ��36.5g+5g+5g+5g-2.2g=49.3g��
���49.3��
��2���ɱ��������ݷ�����֪���ڶ��ν��е�ʵ����ǡ����ȫ��Ӧ��������CO2��������36.5g+5g+5g-44.3g=2.2gʱ���������Ʊ��ʲ�����̼������ȫ��Ӧ��������Ʒ�б��ʲ���̼���Ƶ�����Ϊx��
Na2CO3+2HCl�T2NaCl+H2O+CO2��
106 44
x 2.2g
=
x=5.3g��
2NaOH+CO2�TNa2CO3+H2O
80 106
Y 5.3g
=
y=4g
�ռ���Ʒ���ռ�Ĵ��ȣ�
��100%=60%
���ռ���Ʒ���ռ�Ĵ���Ϊ60%��
���49.3��
��2���ɱ��������ݷ�����֪���ڶ��ν��е�ʵ����ǡ����ȫ��Ӧ��������CO2��������36.5g+5g+5g-44.3g=2.2gʱ���������Ʊ��ʲ�����̼������ȫ��Ӧ��������Ʒ�б��ʲ���̼���Ƶ�����Ϊx��
Na2CO3+2HCl�T2NaCl+H2O+CO2��
106 44
x 2.2g
| 106 |
| 44 |
| x |
| 2.2g |
x=5.3g��
2NaOH+CO2�TNa2CO3+H2O
80 106
Y 5.3g
| 80 |
| y |
| 106 |
| 5.3g |
y=4g
�ռ���Ʒ���ռ�Ĵ��ȣ�
| 10g-4g |
| 10g |
���ռ���Ʒ���ռ�Ĵ���Ϊ60%��
�����������ѶȽϴ�����������������ݣ�

��ϰ��ϵ�д�
�����Ŀ
����˵����ȷ���ǣ�������
| A����ѧ�仯������������������������ |
| B������ȼ�ŵĻ����Һ�������Ƿ�й© |
| C����ˮ���¶ȼƲ�������--�¶����ߣ�ԭ�ӱ�� |
| D����ú¯�Ϸ�һ��ˮ�����Է�ֹһ����̼�ж� |
����˵����ȷ���ǣ�������
| A����һ���ȶ�����Һ��һ������Һ |
| B����ϴ�ྫ�����Ͷ���ϴ�����ۣ��������õ�ԭ����ͬ |
| C��ʳ�Ρ�����ͭ����Һ�ܵ�������Ϊ��Һ���������ƶ������� |
| D�����˲���ʱҪ�ò�������©���в��Ͻ��� |
���з���ֻ������������ǣ�������
| A��Cu |
| B��O2 |
| C��3H2O |
| D��NaCl |
�����ǻ�ѧѧϰ�г��õ�˼ά����������������ȷ���ǣ�������
| A���������ж�������Ԫ�أ����Ժ���Ԫ�صĻ�����һ���������� |
| B��������Һ��ʹ��̪��Һ��죬�����̪��Һ�������Һһ���ʼ��� |
| C����Һ���о���������������������С���������ʵ���������һ����С |
| D�������ɽ������Ӻ����������ɵģ�NH4NO3��û�н������ӣ��������� |
��ȷ�IJ�����ʵ��ɹ�����Ҫ��֤������ʵ������д�����ǣ�������
A�� ���� |
B�� ȡ�����ĩ |
C�� Ϩ��ƾ��� |
D�� ��������� |
���з�Ӧ���ڻ��Ϸ�Ӧ���ǣ�������
A��3Fe+2O2
| ||||
B��CaCO3
| ||||
| C��Zn+2HCl�TZnCl2+H2�� | ||||
| D��Ca��OH��2+CO2�TCaCO3��+H2O |
�������ԭ������Ϊ56��������26�����ӣ�����ԭ�Ӻ��ں��е��������ǣ�������
| A��26 | B��28 | C��30 | D��56 |