��Ŀ����
3��Ϊ�����о����ڷ�����������ҩƷ�����������ȤС���ͬѧ���������ʵ�飬����Ҫʵ�鲽�����£�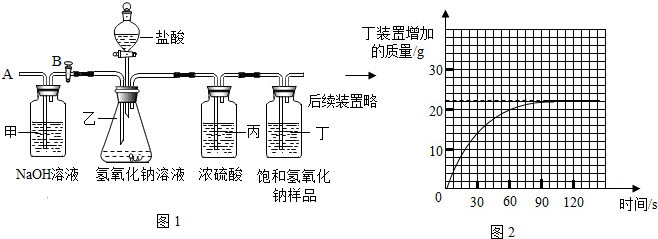
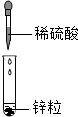
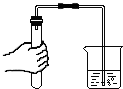
��ͼ1��װ��������55.0g����������ƿ����������ϡ������Һ��
����һ��ʱ����װ�ö����������ӵ���������ͼ2���ߣ�
����ƿ�в��ٲ������ݣ�����B���ӵ���A����������һ�����Ŀ�����
�����ٴγ���װ�ö����������ӵ�������
�����ظ���͢��IJ�����ֱ��װ�ö��������������䣮
�����ʵ�����ݣ�ͨ����������������Ƶı����������ȫ���ʡ��������ֱ��ʡ������ʡ���Ҫ����ϸ�ļ��㲽�裬û�в��費�÷֣�
���� ����װ�ö����������ӵ�����ǡ���Ƕ�����̼�������������ٸ��ݻ�ѧ����ʽ���㼴�ɣ�
��� �⣺��Ϊװ�ö����������ӵ�����ǡ���Ƕ�����̼����������������̼��������22g��
�貿�ֱ��ʵ��������ƹ�����̼���Ƶ�����Ϊx��
Na2CO3+H2SO4�TNa2SO4+H2O+CO2��
106 44
x 22g
$\frac{106}{x}=\frac{44}{22g}$��
��ã�x=53g��
��53g��55g��
�ಿ�ֱ��ʣ�
���� ���⿼�����������Ʊ��ʵ��й�֪ʶ����ɴ��⣬�����������е�֪ʶ���У�Ҫ��ͬѧ����ƽʱ��ѧϰ�м�ǿ����֪ʶ�Ĵ������Ա����Ӧ�ã�

��ϰ��ϵ�д�
 �������Ӧ���⼯ѵϵ�д�
�������Ӧ���⼯ѵϵ�д� �ۺ��Բ�ϵ�д�
�ۺ��Բ�ϵ�д�
�����Ŀ
14���ӷ�������ԾȵȰ�ȫ֪ʶ�жϣ�����������ȷ���ǣ�������
| A�� | �����Ż����Ͽ��Ŵ� | B�� | ����ʱ���Ż��ù��Ǹ��� | ||
| C�� | ���������ȼ���Ƿ�й© | D�� | �����Ż�ʱ��ˮ���� |
18��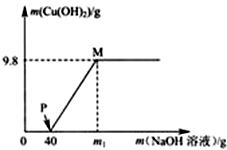 ����CuCl2��HCl�Ļ����Һ100g�У���μ�����������Ϊ10%��NaOH��Һ���μӷ�Ӧ��NaOH��Һ��������������������ϵ��ͼ�������dz���ΪCu��OH��2��������˵����ȷ���ǣ�������
����CuCl2��HCl�Ļ����Һ100g�У���μ�����������Ϊ10%��NaOH��Һ���μӷ�Ӧ��NaOH��Һ��������������������ϵ��ͼ�������dz���ΪCu��OH��2��������˵����ȷ���ǣ�������
 ����CuCl2��HCl�Ļ����Һ100g�У���μ�����������Ϊ10%��NaOH��Һ���μӷ�Ӧ��NaOH��Һ��������������������ϵ��ͼ�������dz���ΪCu��OH��2��������˵����ȷ���ǣ�������
����CuCl2��HCl�Ļ����Һ100g�У���μ�����������Ϊ10%��NaOH��Һ���μӷ�Ӧ��NaOH��Һ��������������������ϵ��ͼ�������dz���ΪCu��OH��2��������˵����ȷ���ǣ�������| A�� | ������Ӧ�����У������Ӹ������� | B�� | P����Һ�е�����ֻ��һ�� | ||
| C�� | M����Һ�����ʵ�����Ϊ15.35g | D�� | ��ȫ����ʱ��m1����ֵΪ80g |
15�������г������ʷ��࣬��ȷ���ǣ�������
��ʳ��ˮ�ڽ���������ʯ�Ң��ռ���������߲�����ʯī��
��ʳ��ˮ�ڽ���������ʯ�Ң��ռ���������߲�����ʯī��
| A�� | ���ڴ�������Ǣڢܢݢޢߢ� | B�� | �ܹ�������Ǣ٢ڢܢݢ� | ||
| C�� | ���ڵ��ʵ��Ǣڢ� | D�� | ���ھ�����Ǣۢܢޢ� |