��Ŀ����
��Ԫ���������ϵ���У�
��1�����ǵؿ��к�����ߵ� Ԫ�أ�
�� 2 �� Ԫ ���� �� ���� �� Ԫ�� �� �� Ϣ�� �� ͼ�� ʾ ���� ԭ �� �ĺ� �� ������Ϊ ��
��3����Ԫ������Ȼ������Ҫ�� Al2 O3 ���ڣ� Al2 O3 ���� ������ĸ��ţ���
A������ B�������� C���� D����
 ��4����ҵ�õ������ Al2 O3 �ķ���ұ��������ѧ����ʽΪ 2Al2 O3
��4����ҵ�õ������ Al2 O3 �ķ���ұ��������ѧ����ʽΪ 2Al2 O3
��Ӧ���ڻ�����Ӧ�����е� ��Ӧ��
��5�������Ƴ���������Ҫ����������չ�Ժ� �ԣ�
ͨ�� ����ʯ
4Al+3O2��,��
��6��ҩƷ����������[ Al(OH)3
]��Ƭ������θ�ᣨ��Ҫ�ɷ�Ϊ���ᣩ�����кͷ�Ӧ����
��θ�����֢���÷�Ӧ�Ļ�ѧ����ʽΪ ��
1������
��2��13
��3��B
��4���ֽ�
��5������
��6�� Al(OH)3 + 3HCl = AlCl3 + 3H2O

Ƥ����һ���й���ͳ��ʳ�����ǽ�Ѽ������һ�����Ƶ���Һ�У��ټӹ����ɡ���������ش��й����⡣
���������ϡ������ʳ�μ��������У��ӷ�ˮ�ܽ⣬������������ʯ�ҳ�ַ�Ӧ����ȴ�õ���Һ��
д����Һ���������з�����Ӧ�Ļ�ѧ����ʽ�� �� ���� �� ��
��������⡿��Һ�г�����NaOH��NaCl�⣬��������Щ���ʣ�
���� �롿
�ף�ֻ��Ca��OH��2��
�ң�ֻ��Na2CO3��
��������Ca��OH��2��Ҳ��Na2CO3��
�����²��У��϶���������� �� ��������������̽����
��ʵ��̽����
ʵ�鲽�� | ʵ������ | ���� |
��ȡ�����ϲ���Һ���ˣ�ȡ��Һ�μ�����ϡ���� | ���������� | �� ��ȷ |
����ȡ������Һ���μ�̼������Һ21�� | ������ɫ���� |
��������˼����
��1����ʯ�Ҽ����ˮ�У�ˮ�ܳ������ַ��ڣ����Ʋ���ԭ������ �� ��
��2��ʵ��ټ�������ϡ�����Ŀ������ �� ��
��3��Ƥ��ʳ��ʱ����һЩʳ�ף�����ȥ��ɬ�У�ԭ������ �� ��

|
| ���� ���� | ��ʵ�� 1���о�ȼ������ | ��ʵ�� 2���о��������� |
| �� | �ձ���ʢ�� 80�����ˮ�� �ֱ���ȼ�ճ��ձ��е� �ܿڷ���һС����ף��� ��ƿ�� | �ձ���ʢ�� NaOH ��Һ�� ȼ�ճ��з���ľ̿ ��ȼľ̿��Ѹ�ٽ�ȼ�� ������ƿ�У�����ƿ�� |
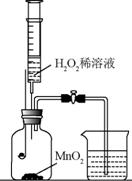
| �� | �������� H2 O2 ��Һ | �������� H2 O2 ��Һ |
��1�� H2 O2 ϡ��Һ�� MnO2 �Ӵ�ʱ������Ӧ�Ļ�ѧ����ʽΪ ��
��2��ʵ�� 1 �У�����H2O2 ��Һǰ��ȼ�ճ�ˮ�еİ�����ȼ�գ�ԭ��ֱ��� �� ���� H2O2��Һ�۲쵽�ձ��е������� ��
��3��ʵ�� 2 �У����� H2 O2 ��Һ�۲쵽ľ̿ȼ�յø����ң��ɴ˵ó����������� �� ��ľ̿Ϩ�����ȴһ��ʱ�䣬�ձ��еIJ�����Һ���뼯��ƿ����ʵ���У�����ƿ��ѹǿ�ı仯������
��

 TiO2��Ϊԭ�����������ѵIJ�����Ҫ�У����ڸ����£�����ʯ�뽹̿�Ļ������ͨ�� ������Cl2�������� TiCl4 ��һ�ֿ�ȼ�����壻����ϡ�����廷���ͼ��������£��� Mg ��TiCl4 ��Ӧ�õ� Ti �� MgCl2������˵����������ȷ������
TiO2��Ϊԭ�����������ѵIJ�����Ҫ�У����ڸ����£�����ʯ�뽹̿�Ļ������ͨ�� ������Cl2�������� TiCl4 ��һ�ֿ�ȼ�����壻����ϡ�����廷���ͼ��������£��� Mg ��TiCl4 ��Ӧ�õ� Ti �� MgCl2������˵����������ȷ������