��Ŀ����
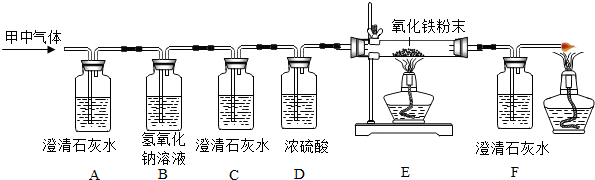
8��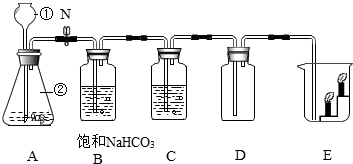 ������ʵ�����Ʊ�CO2ʵ��װ��ͼ����Ҫ��ش��������⣺
������ʵ�����Ʊ�CO2ʵ��װ��ͼ����Ҫ��ش��������⣺��1��д����Ţٵ��������Ƴ���©����ʵ�����Ʊ�CO2�Ļ�ѧ����ʽΪCaCO3+2HCl=CaCl2+H2O+CO2����
��2���ڼ��װ��A�������Բ���ʱ���رջ���N������������עˮ���۲쵽����©������ƿ�ڵ�Һ����ȶ����䣬����©�������γ�Һ����Һ��û���¶˺���Һ�壬�γ�Һ������©����Һ�治���½�������˵��װ�����������ã�
��3������N����A��B��C��D��ȡ���������CO2���壬װ��B�������dz�ȥHCl�����Ȼ��⣩��װ��C��ʢװ����Ũ���ᣨ��ŨH2SO4����
���� ��1�����ݳ��������ش�����̼��ƺ����ᷴӦԭ����д����ʽ��
��2������װ�������Եļ��鷽����������
��3����������ľ�����������
��� �⣺��1����������dz���©����ʵ������ȡ������̼�ô���ʯ��ϡ���ᷴӦ�������Ȼ��ơ�ˮ�Ͷ�����̼����Ӧ����ʽ�ǣ�CaCO3+2HCl=CaCl2+H2O+CO2����
�������©����CaCO3+2HCl=CaCl2+H2O+CO2����
��2���ڼ��װ��A�������Բ���ʱ���رջ���N������������עˮ���۲쵽����©������ƿ�ڵ�Һ����ȶ����䣬����©�������γ�Һ����Һ��û���¶˺���Һ�壬�γ�Һ������©����Һ�治���½�����˵��װ�����������ã��������©������ƿ�ڵ�Һ����ȶ����䣬����©�������γ�Һ����Һ��û���¶˺���Һ�壬�γ�Һ������©����Һ�治���½�����
��3��̼��������Һ��������ȥ������̼�л��е��Ȼ��⣻��Ũ����Գ��Ӻ�Ķ�����̼������и�������ȥHCl�����Ȼ��⣩��Ũ���ᣨ��ŨH2SO4����
���� ������Ҫ���鳣������ķ���װ�����ռ�װ�õ�̽��������װ�����ݷ�Ӧ���״̬�ͷ�Ӧ����ѡ���ռ�װ������������ܶȺ��ܽ���ѡ��

��ϰ��ϵ�д�
�����Ŀ
2���������Լ���ȥһ����̼��������̼��ˮ��������ʾ��Ũ��������ˮ�ԣ������в���˳����ȷ���ǣ�������
��Ũ���� ��ͨ�����ȵ�����ͭ �۳���ʯ��ˮ��
��Ũ���� ��ͨ�����ȵ�����ͭ �۳���ʯ��ˮ��
| A�� | �٢ڢ� | B�� | �ڢ٢� | C�� | �ڢۢ� | D�� | �ۢ٢� |
19�������ijָʾ����һ��pH��Χ����ʾ����ɫ�����������ָʾ����ϡ�����м�������������Һ�����ֵ���ɫ�仯�����ܵ��ǣ�������
| ��ɫ | �� | �� | �� | �� | �� |
| pH��Χ | 1��3 | 4��5 | 6��7 | 8��10 | 11��14 |
| A�� | �ɺ���� | B�� | �������� | C�� | �ɳȱ��� | D�� | �ɺ���� |
20���й���������Ϊ��ף��սʤ��70���귢���˼���ң�ѡ���������ҵĽ�������ʱ���迼�Ǹý������ϵģ�������
| A�� | ���� | B�� | ������ | C�� | ��ʴ�� | D�� | ������ |
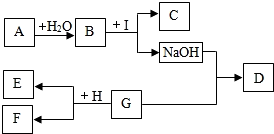 A-IΪ���꼶��ѧ��ѧ�����ʣ���֪AΪ���������CΪ����ʯ����Ҫ�ɷ֣�DΪ��ɫ������EΪ���ʣ�FΪdz��ɫ��Һ��F��G��I��Ϊֻ����һ�����ʵ���Һ����������ʾ���ʼ�������ת���Ĺ�ϵ������������ĩ���������ͼ��ʾ����ش��������⣺
A-IΪ���꼶��ѧ��ѧ�����ʣ���֪AΪ���������CΪ����ʯ����Ҫ�ɷ֣�DΪ��ɫ������EΪ���ʣ�FΪdz��ɫ��Һ��F��G��I��Ϊֻ����һ�����ʵ���Һ����������ʾ���ʼ�������ת���Ĺ�ϵ������������ĩ���������ͼ��ʾ����ش��������⣺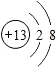 ��
��