��Ŀ����
10�������������������ʵ�ȥ������ѧ������ʵ��֮һ��������ͼ����������⣺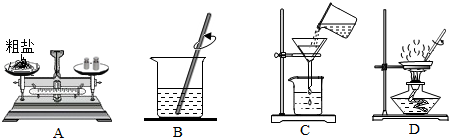
��1���ڲ���A����������ƽ��ȡ5.0g���Σ�Ϊʹ���γ���ܽ⣬����ˮ�����ʹ����ԼΪ13.9mL������������һλС��������֪20��ʱNaCl���ܽ��Ϊ36g����
��2����ͬѧ������C���й��ˣ���ָ��������������δ�ò�����������©���¶˼���δ�����ձ��ڱڣ�
��3���ڲ���D���ȹ����У��ò��������Ͻ��裬��ֹ��ֲ��¶ȹ��ߣ����Һ�ηɽ������������г��ֽ϶����ʱ��ֹͣ���ȣ�
��4��ʵ������������õľ��Σ������㾫�εIJ��ʣ����ֲ���ƫ�ͣ������ԭ����AD������ĸ����
A��ʳ��û��ȫ���ܽ⼴���� B����ֽû�н���©���ڱ�
C�����������þ��κܳ�ʪ D��������մ�еľ���û��ȫ��ת�Ƶ�����ֽ�ϣ�
���� ��1������20��ʱNaCl���ܽ��Ϊ36g������5g����ȫ�����Ȼ��ƣ��������Ҫ��ˮ���������з�����
��2�����ݹ��˲�������Ҫ�ò�����������©���¶˼���Ҫ�����ձ��ڱڽ��з�����
��3���������������в����������ý��з�����
��4�����ݴ����ᴿ�����Ĺ��̼�ע���������з�����
��� �⣺��1��20��ʱNaCl���ܽ��Ϊ36g������5g����ȫ�����Ȼ��ƣ�
����ҪˮΪx
100g��36g=x��5g
x=13.9g��13.9mL��
��2�����˲�������Ҫ�ò�����������©���¶˼���Ҫ�����ձ��ڱڣ�������ͬѧ������C���й��ˣ��������������ǣ�δ�ò�����������©���¶˼���δ�����ձ��ڱڣ�
��3�������������У��ò��������Ͻ��裬��ֹ��ֲ��¶ȹ��ߣ����Һ�ηɽ������������г��ֽ϶����ʱ��ֹͣ���ȣ�
��4��A��ʳ��û��ȫ���ܽ⼴���ˣ�����ʧһ�����Ȼ��ƣ����Իᵼ�²��ʽ��ͣ���A��ȷ��
B����ֽû�н���©���ڱڣ���ʹ�����ٶȼ��������ᵼ�²��ʽ��ͣ���B����
C������ʱ���õ�ʳ�γ�ʪ��������ں�����ˮ�֣������²���ƫ��C����
D��������մ�еľ���ûȫ��ת�Ƶ�����ֽ�ϣ���ʹ��õľ��μ��٣���ʹ����ƫ�ͣ���D��ȷ��
��ѡ��AD��
�ʴ�Ϊ����1��13.9��
��2��δ�ò�����������©���¶˼���δ�����ձ��ڱڣ�
��3����ֲ��¶ȹ��ߣ����Һ�ηɽ����϶���壻
��4��AD��
���� �����ۺϿ����˴����ᴿʵ����й�ע��������ʱҪ�������֪ʶϸ�ķ�����

ʵ����� ��� | ʵ��Ŀ�� | �����Լ���������� | ʵ������ | ʵ����� |
| A | ����ij����������Ƿ���CO2���� | ͨ�����ʯ��ˮ | ���������� | �������в���CO2 |
| B | ��ȥKCI�е�����K2CO3 | ϡ���� | �����ݲ��� | K2CO3�ѱ����� |
| C | ����Ӳˮ������ˮ | ����ˮ | ����������ĭ | ��Һ��Ϊ����ˮ |
| D | ���ſ������ռ�CO2ʱ���鼯��ƿ��CO2�Ƿ��� | ��ȼ�յ�ľ������ƿ�� | ľ��Ϩ�� | ƿ��CO2�Ѽ��� |
| A�� | A | B�� | B | C�� | C | D�� | D |
| A�� | �ƾ��ӷ� | B�� | �������� | C�� | ��ˮɹ�� | D�� | �ɱ����� |
| ѡ�� | ʵ������ | ʵ�鷽�� |
| A | ����ij�����������Ƿ���̼���� | ȡ������Ʒ���Թ��У��μ�������ϡ���ᣬ���������ݲ�������ù��������к���̼���� |
| B | ����CO��CO2 | �ֱ�����ƿ�м���ˮ�����ܽ���Ƕ�����̼ |
| C | ��ȥCO2�л��е�CO | ���������ͨ������������������Һ���ٽ�һ����̼��ȼ��ȥ |
| D | ����̼���ƺ�̼������ֹ����ĩ | ȡһ�ֹ����������Թ��У�������ˮ�����������ܽ⣬������̼���ƣ������ܽ⣬������̼��� |
| A�� | A | B�� | B | C�� | C | D�� | D |
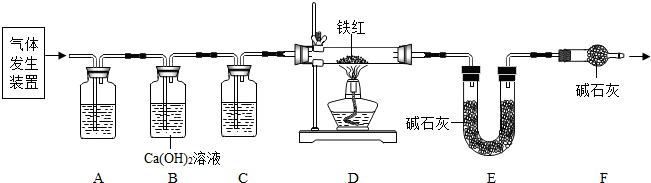
 ��ͼ�����Ǿ���ʹ�õĽ�ͨ�������г�����������и��⣮
��ͼ�����Ǿ���ʹ�õĽ�ͨ�������г�����������и��⣮