��Ŀ����
ij��ѧ��ȤС�齫�����ͬ����
��������ͬ����,�ֱ�����ݲ�ͬ���������
�ƾ���Һ�н���ȡ����𣬽����ˡ��ղ���
����������ʵ�顣��ʵ�������±���
| ʵ���顡�� | �� | �� | �� | �� | �� |
| �ƾ��������/% | 95 | 65 | 55 | 15 | 5 |
| ʵ���顡�֡��� | �ƾ����� �����ջ� | �ƾ����� �������� | �ƾ����� �������� | ��ȼ�� | �� |
�������Ʋ�ݵ�ʵ��������������������������
�����ۺ���ѧ֪ʶ���ڡ����С��ƾ�������������𡱵�ԭ������������������������������������
�Ų�ȼ�գ��Ǿƾ��������ȣ�ʹ�������¶ȵ����Ż�㡣

���л�ѧ����ʽ��д��ȫ��ȷ���ǣ�
|
|
|
|
C. 4P+5O2 2P2O5 D. 2C2H2+5O2====4CO2+2H2O
Ԫ�����ڱ���ѧϰ���о���ѧ����Ҫ���ߡ�������±���Ԫ�����ڱ��IJ������ݣ��ش��й����⣺
|
���� | I A | II A | IIIA | IVA | VA | VIA | VIIA | 0 |
| 2 | 3 Li � 6.941 | 4 Be �� 9.012 | 5 B �� 10.81 | 6 C ̼ 12.01 | 7 N �� 14.01 | 8 O �� 16.00 | 9 F �� 19.00 | 10 Ne �� 20.18 |
| 3 | 11 Na �� 22.99 | 12 Mg þ 24.31 | 13 Al �� 26.98 | 14 Si �� 28.09 | 15 P �� 30.97 | 16 S �� 32.06 | 17 Cl �� 35.45 | 18 Ar � 39.95 |
��1������ϱ��в�����ڹ�Ԫ�ص�һ����Ϣ��___________________________��
��2����3���ڣ����У������ڽ���Ԫ�ص��� ����һ�֣����������ӵķ��� Ϊ ��
Ϊ ��
��3����11��Ԫ�����17��Ԫ����ɵĻ������� �����ɸ����ʵ����� ��ѡ����ӡ�����ԭ�ӡ������ӡ�����
��4����Ԫ�����ڱ��У�ͬһ�壨���У���Ԫ�ؾ������ƵĻ�ѧ���ʡ������и���Ԫ�ؾ������ƻ�ѧ���ʵ��� ������ţ���
a. C��Ne b. Be��Mg c. Al��Si d. F��Cl
��5����9�ŷ�Ԫ������Ԫ���γɵĻ������ˮ��Һ����ᣨHF�������ڲ������, ����Ҫԭ����������벣������Ҫ�ɷֶ������裨SiO2��������Ӧ�������ķ��������壨SiF4����ˮ���÷�Ӧ�Ļ�ѧ����ʽΪ ��
����Ҫ��д���±���Ӧ�Ļ�ѧ���š�
| ������ | ��ԭ�� | ���������� | ��������� | ��ʯ�� | ����þ���� |
| ��ѧ���� |
 ��
�� Cu+CO2��
Cu+CO2��  B��Cl2+H2O=HCl+HClO��
B��Cl2+H2O=HCl+HClO��  H2+CO��
H2+CO��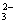
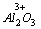 D.4����ԭ�ӣ�2H2
D.4����ԭ�ӣ�2H2