��Ŀ����
�ֿ�����һ�����ʵİ�װ��ǩģ�����壬��ͬѧ����������̽����
��1���������ò��������Ŀ���� ��
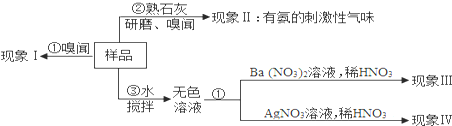
��2��������±�����֪AgCl��BaSO4��������ˮ����İ�ɫ���壩��
��3�������������������õ��ʿ����� ��д��ѧʽ����������������Ϊ����ζ���������������õ��ʿ����� ��ֻдһ�֣���
��1���������ò��������Ŀ����

��2��������±�����֪AgCl��BaSO4��������ˮ����İ�ɫ���壩��
| ��������� | �жϺͻ�ѧ����ʽ | |
| a | ������IΪ�ް�ζ�� | ����Ʒ�в��� |
| b | �ɲ����������жϣ� | ����Ʒһ������ |
| c | �������Ϊ��ɫ�����������Ϊ������ | ����Ʒ�к��� |
| d | �������Ϊ���������������Ϊ��ɫ������ | ����Ʒ�к��� |
���㣺ȱʧ��ǩ��ҩƷ�ɷֵ�̽��,�εĻ�ѧ����,�̬���ʵļ���,�������ӵļ��鷽��������,��д��ѧ����ʽ�����ֱ���ʽ�����뷽��ʽ
ר�⣺��ѧ̽��
��������1�����ݲ����������÷�����
��2��a�����ݳ�����̼������ֽ�����ʷ�����
b������笠����ӵļ��鷽��������
c��������������ӵļ��鷽��������
d�����������ӵļ��鷽��������
��3�����������ε����ʡ������ĵ��ʵ����������
��2��a�����ݳ�����̼������ֽ�����ʷ�����
b������笠����ӵļ��鷽��������
c��������������ӵļ��鷽��������
d�����������ӵļ��鷽��������
��3�����������ε����ʡ������ĵ��ʵ����������
����⣺��1���������ò��������Ŀ����ʹ��Ʒ����ܽ⣻
��2��a�����ڳ�����̼������ֽ�ų��������д̼�����ζ�����ԣ�������IΪ�ް�ζ������Ʒ�в���̼����泥�
b��������������ܷų����������ԣ�����Ʒ������ʯ�ң��а������ɣ�˵���˸���Ʒһ������笠����ӣ��õ���������Σ�
C��������������ᱵ��Һ��Ӧ���ɲ�����ϡ����İ�ɫ������˵���˸����������泥���Ӧ�ķ���ʽ����NH4��2SO4+Ba��NO3��2=BaSO4��+2NH4NO3��
d�������������������Һ��Ӧ���ɲ�����ϡ����İ�ɫ������˵���˸�������Ȼ�泥���Ӧ�ķ���ʽ��NH4Cl+AgNO3=AgCl��+NH4NO3��
��3�������������������õ��ʿ����� NH4NO3��������������Ϊ����ζ���������������õ��ʲ�����Σ����������أ�
�ʴ�Ϊ����1��ʹ��Ʒ����ܽ⣻��2��a��̼����泥�b��笠���c������泥���NH4��2SO4+Ba��NO3��2=BaSO4��+2NH4NO3��d��NH4Cl NH4Cl+AgNO3=AgCl��+NH4NO3����3��NH4NO3�����أ�
��2��a�����ڳ�����̼������ֽ�ų��������д̼�����ζ�����ԣ�������IΪ�ް�ζ������Ʒ�в���̼����泥�
b��������������ܷų����������ԣ�����Ʒ������ʯ�ң��а������ɣ�˵���˸���Ʒһ������笠����ӣ��õ���������Σ�
C��������������ᱵ��Һ��Ӧ���ɲ�����ϡ����İ�ɫ������˵���˸����������泥���Ӧ�ķ���ʽ����NH4��2SO4+Ba��NO3��2=BaSO4��+2NH4NO3��
d�������������������Һ��Ӧ���ɲ�����ϡ����İ�ɫ������˵���˸�������Ȼ�泥���Ӧ�ķ���ʽ��NH4Cl+AgNO3=AgCl��+NH4NO3��
��3�������������������õ��ʿ����� NH4NO3��������������Ϊ����ζ���������������õ��ʲ�����Σ����������أ�
�ʴ�Ϊ����1��ʹ��Ʒ����ܽ⣻��2��a��̼����泥�b��笠���c������泥���NH4��2SO4+Ba��NO3��2=BaSO4��+2NH4NO3��d��NH4Cl NH4Cl+AgNO3=AgCl��+NH4NO3����3��NH4NO3�����أ�
�����������Ĺؼ��������̼����淋����ʡ�笠����ӡ���������ӡ������Ӽ��鷽����֪ʶ��

��ϰ��ϵ�д�
�����Ŀ
 ��ͼ��ʾ��װ����ʵ�������ж�����;�����ڸ�װ�õ�˵ ���в���ȷ���ǣ�������
��ͼ��ʾ��װ����ʵ�������ж�����;�����ڸ�װ�õ�˵ ���в���ȷ���ǣ�������| A����װ����ʵ�����п���������ȡ���� |
| B����װ��Ҳ������ʵ������ȡ������̼ |
| C����װ����ȡ�����巴Ӧ���ǹ����Һ�壬����Ҫ���� |
| D����װ����ʵ�����п�����������ƿ�е����塰����������ƿ�� |
����ͼ������ȷ��ӳ��Ӧ�仯��ϵ���ǣ�������
A�� ��һ�����Ķ��������м������������Һ |
B��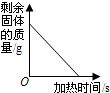 ���ȸ��������ȡ���� |
C��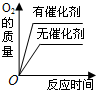 �õ���������Ũ�ȵĹ���������Һ�ֱ���ȡ���� |
D�� ��ˮͨ��ֽ�һ��ʱ�� |
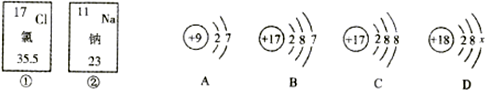
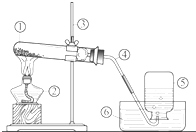 ��ͼ��ʵ�����ø��������ȡ������װ�ã�
��ͼ��ʵ�����ø��������ȡ������װ�ã� ���ִ������У�����Խ��Խע����Ԫ�ص���ȡ����Ԫ�ض���������������Ҫ�����ã���ͼ��ij���г����۵�һ�֡��ӵ�ʳ�Ρ���װ���ϵIJ���˵������ش��������⣺
���ִ������У�����Խ��Խע����Ԫ�ص���ȡ����Ԫ�ض���������������Ҫ�����ã���ͼ��ij���г����۵�һ�֡��ӵ�ʳ�Ρ���װ���ϵIJ���˵������ش��������⣺