��Ŀ����
�ݱ�����̫���ܹ���������Ϻ��������ϵõ��˴��ģ��Ӧ�ã���װ����������4.68���ߡ�
��1����չ̫���ܲ�ҵ��Ҫ�ŵ��ǣ���һ�㼴�ɣ�___________________��
��2��̫���ܹ��������ؼ��IJ����Ǹߴ��裬��ͼ������ij�ྦྷ��������ҵ�����ߴ��������ʾ��ͼ��
��1����չ̫���ܲ�ҵ��Ҫ�ŵ��ǣ���һ�㼴�ɣ�___________________��
��2��̫���ܹ��������ؼ��IJ����Ǹߴ��裬��ͼ������ij�ྦྷ��������ҵ�����ߴ��������ʾ��ͼ��
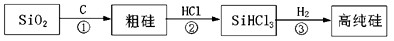
�����ķ�ӦΪ����SiO2+2C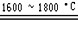 Si+2CO������Si+3HCl
Si+2CO������Si+3HCl SiHCl3+H2������SiHCl3+H2
SiHCl3+H2������SiHCl3+H2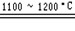 Si+3HCl�������Ʊ����̱���ﵽ��ˮ��������H2��ԭSiHCl3������������O2����������ĺ����________________��
Si+3HCl�������Ʊ����̱���ﵽ��ˮ��������H2��ԭSiHCl3������������O2����������ĺ����________________��
��3��Ϊ�˴ﵽ��ɫ��ѧ�ͽ�Լ��Դ��Ŀ�ģ�����������ij��������Ҫѭ��ʹ�ã������ʵĻ�ѧʽ��________��
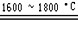 Si+2CO������Si+3HCl
Si+2CO������Si+3HCl SiHCl3+H2������SiHCl3+H2
SiHCl3+H2������SiHCl3+H2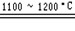 Si+3HCl�������Ʊ����̱���ﵽ��ˮ��������H2��ԭSiHCl3������������O2����������ĺ����________________��
Si+3HCl�������Ʊ����̱���ﵽ��ˮ��������H2��ԭSiHCl3������������O2����������ĺ����________________����3��Ϊ�˴ﵽ��ɫ��ѧ�ͽ�Լ��Դ��Ŀ�ģ�����������ij��������Ҫѭ��ʹ�ã������ʵĻ�ѧʽ��________��
��1������Ⱦ�����ڻ���������̼�ŷš����ٻ�ʯ��Դ���ĵȣ�
��2����ը
��3��HCl����H2��
��2����ը
��3��HCl����H2��

��ϰ��ϵ�д�
 ��ѧ�̸̳����¿α�ϵ�д�
��ѧ�̸̳����¿α�ϵ�д� Сѧ��ʱ��ѵϵ�д�
Сѧ��ʱ��ѵϵ�д�
�����Ŀ

 SiHCl3+H2��
SiHCl3+H2�� Si+3HCl
Si+3HCl
 Si+2CO��
Si+2CO�� SiHCl3+H2��
SiHCl3+H2�� Si+3HCl
Si+3HCl
 Si+2CO��
Si+2CO�� SiHCl3+H2��
SiHCl3+H2�� Si+3HCl
Si+3HCl
 Si+2CO��
Si+2CO�� SiHCl3+H2��
SiHCl3+H2�� Si+3HCl
Si+3HCl