��Ŀ����
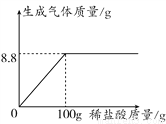
�ں�����NaCl��ij������Ʒ23g�м������ᣬ��Ӧ�в���������������������������ϵ��ͼ��ʾ�����㵱ǡ����ȫ��Ӧʱ��
(1)�����Ķ�����̼�������ǡ�__________��g��
(2)�˴�����Ʒ��̼���Ƶ���������(д����Ҫ�ļ�����̣����������0.1%)��_________.
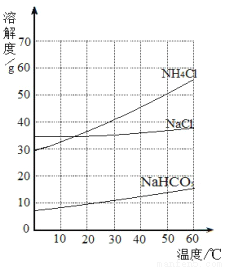
��ϰ��ϵ�д�
 ���Ͱ�ͨ��ĩ���ϵ�д�
���Ͱ�ͨ��ĩ���ϵ�д�
�����Ŀ
���г�ȥ����(����)�ķ�������ȷ����
ѡ�� | ���� | ���� | ���ӷ��� |
A | CaCO3���� | CaO | ������ϡ���ᣬ���� |
B | HCl���� | H2O | ͨ���������ƹ��壬�ռ� |
C | NaOH��Һ | Na2CO3 | ����������ʯ��ˮ������ |
D | MnO2 | NaCl���� | �ܽ⡢���ˡ�ϴ�ӡ���� |
A. A B. B C. C D. D
D ��������A��CaO��CaCO3������ϡ���ᷴӦ�������ܰ����ʳ�ȥ��Ҳ���ԭ���ʳ�ȥ�������ϳ���ԭ����B�������������ڼ������ʣ��������������������壬����C������ʯ��ˮ�ܰ�̼���Ƴ�ȥ�����������µ������������ƣ�����D���������̲�����ˮ���Ȼ���������ˮ���ʿ����ܽ⡢���ˡ�ϴ�ӡ������������ӣ���ȷ����ѡD��






 Һ����㵹 B.
Һ����㵹 B. 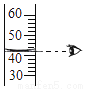 Һ�����ȡ
Һ�����ȡ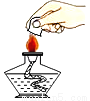 Ϩ��ƾ��� D.
Ϩ��ƾ��� D. 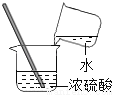 ϡ��Ũ����
ϡ��Ũ����