��Ŀ����
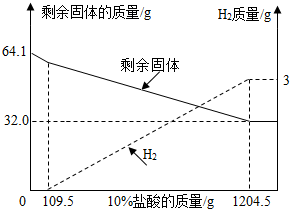 С��ͬѧ���ڿ����з���һ��ʱ��ġ�ͭ��������˿����������ͭ��Һ���Ƴɣ���ͼ���ijɷֽ��з�����ȡ��64.1g������Ʒ����10%���������ܽ⣬��Һ����ɫ�������������ʣ�����������10%�����������仯��ϵ������ͼ��
С��ͬѧ���ڿ����з���һ��ʱ��ġ�ͭ��������˿����������ͭ��Һ���Ƴɣ���ͼ���ijɷֽ��з�����ȡ��64.1g������Ʒ����10%���������ܽ⣬��Һ����ɫ�������������ʣ�����������10%�����������仯��ϵ������ͼ�����ͼ�����ݷ�����
��1����ͼ��֪��CuԪ�ص�����
��2�����ϻ�ѧ����ʽ����á�ͭ������AlԪ�ص�����������
��3��ֻ֪����ͭ��������m1������10%��������������m2����Ҳ�������ͭ��������Ԫ�ص����������������ʽΪ
���㣺���ݻ�ѧ��Ӧ����ʽ�ļ���
ר�⣺�ۺϼ��㣨ͼ���͡������͡��龰�ͼ����⣩
���������ݼ������ᷴӦ�������������仯����ֵ��֪ͭ�����������ݷ�Ӧ��ʼʱ����������С�����������������ص����ͩ���ijɷ֣�����ͩ����ͭ���ʺ���������ͭԪ�ص���������ͩ������Ԫ�ص��������������ݷ�Ӧ�����ɵ��Ȼ�������Ԫ�ص����������������Ԫ�ص�����������
����⣺��1������ͩ����ֻ��ͭ���������ᷴӦ�����Լ������������������ټ�С�Ĺ���������Ϊͭ������������ֵΪ32.0g��
����ͼ���м�������109.5gǰ����������С��������������������ͭ���������ᷴӦ�������ܷ�Ӧ������������������ͩ����ֻ�ܴ�����������Al2O3����
��2����ͼ֪����H2������Ϊ3g���衰ͭ������Al���ʵ�����Ϊx��
2Al+6HCl�T2AlCl3+3H2��
54������������������6
x������������������3 g
=
���x=27g
����ͭ������Al���ʵ�����Ϊ27g��
��ͭ������Al2O3������Ϊ64.1g-27g-32g=5.1g����Al2O3�к�AlԪ�ص�����Ϊ2.7 g�����Ըá�ͭ�����к�AlԪ�ص���������Ϊ
��100%=46.3%��
��3���������ᷴӦ����Ԫ��ȫ��תΪ�Ȼ����е���Ԫ�أ����Ծݴ˿ɼ������Ԫ�ص�������
����Ԫ�ص�����Ϊy
����
=
y=
��ͭ��������Ԫ�ص���������Ϊ
��100%��
�ʴ�Ϊ����1��32��Al2O3����2���á�ͭ������AlԪ�ص���������Ϊ46.3%����3��
��100%��
����ͼ���м�������109.5gǰ����������С��������������������ͭ���������ᷴӦ�������ܷ�Ӧ������������������ͩ����ֻ�ܴ�����������Al2O3����
��2����ͼ֪����H2������Ϊ3g���衰ͭ������Al���ʵ�����Ϊx��
2Al+6HCl�T2AlCl3+3H2��
54������������������6
x������������������3 g
| 54 |
| x |
| 6 |
| 3g |
���x=27g
����ͭ������Al���ʵ�����Ϊ27g��
��ͭ������Al2O3������Ϊ64.1g-27g-32g=5.1g����Al2O3�к�AlԪ�ص�����Ϊ2.7 g�����Ըá�ͭ�����к�AlԪ�ص���������Ϊ
| (27g+2.7g) |
| 64.1g |
��3���������ᷴӦ����Ԫ��ȫ��תΪ�Ȼ����е���Ԫ�أ����Ծݴ˿ɼ������Ԫ�ص�������
����Ԫ�ص�����Ϊy
����
| y | ||
m2��10%��
|
| 27 |
| 106.5 |
y=
27��m2��10%��
| ||
| 106.5 |
��ͭ��������Ԫ�ص���������Ϊ
| 9��m2��10% |
| 36.5gm1 |
�ʴ�Ϊ����1��32��Al2O3����2���á�ͭ������AlԪ�ص���������Ϊ46.3%����3��
| 9��m2��10% |
| 36.5gm1 |
�����������ǻ�ѧ����ʽ����Ŀ����⣬����Ĺؼ����ܹ�����ͼ�����صķ�Ӧ���̵������Ӧ�ã�

��ϰ��ϵ�д�
 Сѧ��ʱ��ҵȫͨ����ϵ�д�
Сѧ��ʱ��ҵȫͨ����ϵ�д� �����ÿ�ʱѵ��ϵ�д�
�����ÿ�ʱѵ��ϵ�д�
�����Ŀ
�������һ�㲻����ɿ�����Ⱦ���ǣ�������
| A�������ų���β�� |
| B��ȼ�ղ��������� |
| C��ֲ�������÷ų����� |
| D�����������������̳� |
��ȥ�Ȼ����е�����̼���ƣ����з�������ʵ�ֵ��ǣ�������
| A�����������ᣬ�����ᾧ |
| B���������Ȼ�����Һ�����˺������ᾧ |
| C���������Ȼ�����Һ�����˺������ᾧ |
| D������������������Һ�����˺������ᾧ |
Ҫ������б仯������ͨ�����ᾭһ����Ӧʵ�ֵ��ǣ�������
| A��CaO��CaCl2 |
| B��Fe2O3��FeCl3 |
| C��Cu��CuCl2 |
| D��Fe��FeCl2 |