��Ŀ����
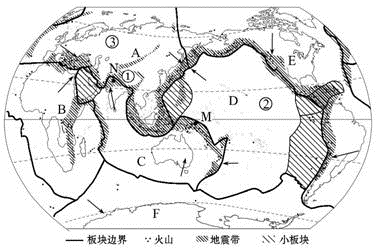
 ������������ͼ���ش��������⣮
������������ͼ���ش��������⣮��1��д��ͼ����ĸ�������İ�����
A
D
��2���������У�������λ�ں����е���
��3��ͼ�Тٴ���λ��
��4�����к�-ϲ�����Ż�ɽ�����������ŷ�����
��5���ҹ�����
���㣺�����鼰��ʾ��ͼ,��ɽ������ֲ������˶��Ĺ�ϵ
ר�⣺
����������ѧ�Ҿ������ڵ��о�������˰�鹹��ѧ˵����Ϊȫ������������ɣ�һ����˵����ڲ��Ƚ��ȶ���������齻��ش����ؿDZȽϻ�Ծ�����ɽ���������������������ŷ��顢���ް�顢ӡ�����顢̫ƽ���顢���ް�顢�ϼ��ް�飮
����⣺��1����ͼ�ɵã�A����ŷ��飬B�Ƿ��ް�飬C��ӡ�����飬D��̫ƽ���飬E�����ް�飬G���ϼ��ް�飮
��2���������У�������λ�ں����е���̫ƽ���飬�Ĵ����Ǵ�½λ��ӡ�������ϣ��ϼ���λ���ϼ��ް���ϣ�
��3��ͼ�Тٴ���λ�ڻ�̫ƽ���ɽ����ش��ϣ��û�ɽ������ش���ŷ����ӡ�����顢̫ƽ���顢���ް�顢�ϼ��ް��Ľ��紦���ڴ���λ�ڵ��к�-ϲ�����Ż�ɽ����ش��ϣ��û�ɽ�����λ����ŷ����ӡ�����顢���ް�齻�紦��
��4�����к�-ϲ�����Ż�ɽ�����������ŷ�����ӡ������ͷ��ް��Ľ���ش���
��5���ҹ�������ŷ��顢ӡ������� ̫ƽ����Ľ���ش��������һ�������ΪƵ���Ĺ��ң�
�ʴ�Ϊ��
��1����ŷ��飻���ް�飻ӡ�����飻̫ƽ���飻���ް�飻�ϼ��ް�飻
��2��̫ƽ���飻ӡ�����ϼ��ޣ�
��3����̫ƽ����ŷ��ӡ����̫ƽ�����ް����ϼ��ޣ����к�-ϲ�����ţ���ŷ��ӡ�����ޣ�
��4��ӡ�����ޣ�
��5����ŷ��ӡ����̫ƽ��
��2���������У�������λ�ں����е���̫ƽ���飬�Ĵ����Ǵ�½λ��ӡ�������ϣ��ϼ���λ���ϼ��ް���ϣ�
��3��ͼ�Тٴ���λ�ڻ�̫ƽ���ɽ����ش��ϣ��û�ɽ������ش���ŷ����ӡ�����顢̫ƽ���顢���ް�顢�ϼ��ް��Ľ��紦���ڴ���λ�ڵ��к�-ϲ�����Ż�ɽ����ش��ϣ��û�ɽ�����λ����ŷ����ӡ�����顢���ް�齻�紦��
��4�����к�-ϲ�����Ż�ɽ�����������ŷ�����ӡ������ͷ��ް��Ľ���ش���
��5���ҹ�������ŷ��顢ӡ������� ̫ƽ����Ľ���ش��������һ�������ΪƵ���Ĺ��ң�
�ʴ�Ϊ��
��1����ŷ��飻���ް�飻ӡ�����飻̫ƽ���飻���ް�飻�ϼ��ް�飻
��2��̫ƽ���飻ӡ�����ϼ��ޣ�
��3����̫ƽ����ŷ��ӡ����̫ƽ�����ް����ϼ��ޣ����к�-ϲ�����ţ���ŷ��ӡ�����ޣ�
��4��ӡ�����ޣ�
��5����ŷ��ӡ����̫ƽ��
���������⿼��������ֲ�ͼ����ͼ��ɣ�

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ
������������ϵ���У����������������������ɲ���Ӱ�죨������
| A����̼���� |
| B����ɫ���� |
| C��ֲ������ |
| D���Ӵ�ߺ��ܲ�ҵ��չ |
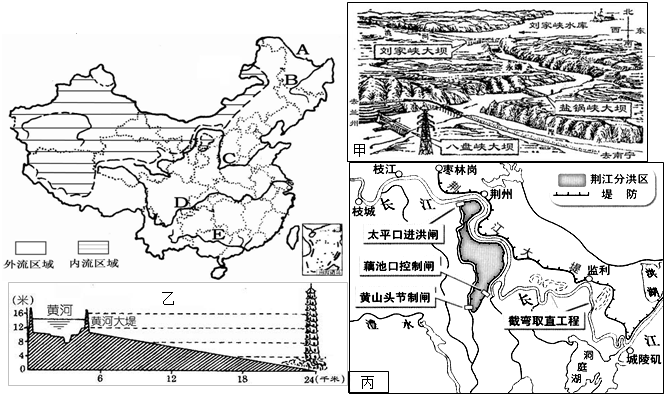
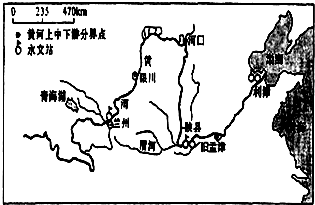 �Ķ����в��ϣ�̽���ƺ���ɳ��������ԭ��
�Ķ����в��ϣ�̽���ƺ���ɳ��������ԭ��