题目内容
.对于一定质量的理想气体,有两个状态A(p1,V1)、B(p2,V2),如图所示.它们对应的温度分别为T1、T2.且T1≠T2,图中的曲线为等温线.请你利用理想气体实验定律证明: ,要求在图上标出与证明过程相应的状态变化图线.
,要求在图上标出与证明过程相应的状态变化图线. 
 ,要求在图上标出与证明过程相应的状态变化图线.
,要求在图上标出与证明过程相应的状态变化图线. 
P1V1/T1= P2 V2/T2
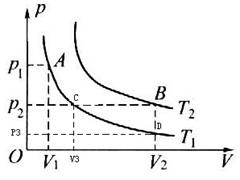
由状态A到状态C遵循玻意耳定律
P1V1 = P2V3------------------------------------------------①
由状态C到状态B遵循盖-吕萨克定律
V3/T1=V2/T2------------------------------------------------②
①式除以T1与②式乘以P2联立得
P1V1/T1= P2 V2/T2

P1V1 = P2V3------------------------------------------------①
由状态C到状态B遵循盖-吕萨克定律
V3/T1=V2/T2------------------------------------------------②
①式除以T1与②式乘以P2联立得
P1V1/T1= P2 V2/T2


练习册系列答案
 海淀黄冈名师导航系列答案
海淀黄冈名师导航系列答案 普通高中同步练习册系列答案
普通高中同步练习册系列答案
相关题目

 ,根据这些条件可求出油分子的直径为__________,
,根据这些条件可求出油分子的直径为__________, ;若等温膨胀需吸收热量
;若等温膨胀需吸收热量 ,则
,则 
 ,由10℃升到20℃压强增加量为△
,由10℃升到20℃压强增加量为△ ,则
,则 



