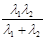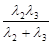题目内容
(6分)如图为氢原子的能级示意图,锌的逸出功是3.34 eV,那么对氢原子在能级跃迁过程中发射或吸收光子的特征认识正确的是 。(填正确答案标号。选对1个得3分,选对2个得4分,
选对3个得6分。每选错1个扣3分,最低得分为0分)
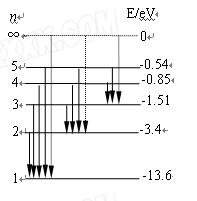
E.用能量为14.0 eV的光子照射,可使处于基态的氢原子电离
选对3个得6分。每选错1个扣3分,最低得分为0分)

| A.用氢原子从高能级向基态跃迁时发射的光照射锌板一定不能产生光电效应 |
| B.一群处于n=3能级的氢原子向基态跃迁时,能放出3种不同频率的光 |
| C.一群处于n=3能级的氢原子向基态跃迁时,发出的光照射锌板,锌板表面所发出的光电子的最大初动能为8.75 eV |
| D.用能量为10.3 eV的光子照射,可使处于基态的氢原子跃迁到激发态 |
BCE
试题分析:氢原子从高能级向基态跃迁时辐射的光子能量等于能级差,只有能级差大于等于锌板逸出功
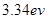 ,比如从
,比如从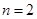 跃迁到基态时,辐射处的光子能量就大于
跃迁到基态时,辐射处的光子能量就大于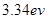 ,可以使锌板发生光电效应,选项A错。一群处于n=3能级的氢原子向基态跃迁时,存在
,可以使锌板发生光电效应,选项A错。一群处于n=3能级的氢原子向基态跃迁时,存在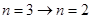 ,
,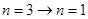 ,
,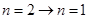 三条路径,辐射出三种不同能量即不同频率的光子,选项B对。辐射出的光子能量等于能极差,逸出光电子的最大初动能
三条路径,辐射出三种不同能量即不同频率的光子,选项B对。辐射出的光子能量等于能极差,逸出光电子的最大初动能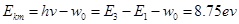 ,选项C对。用能量为10.3 eV的光子照射,由于该光子能量不等于任意两个氢原子的能极差,而且小于从基态到电离所需的能量,所以基态的氢原子不能吸收该光子跃迁,而14.0 eV的光子足以使得氢原子从基态电离,多余的能量转化为电离后的动能,选项D错E对。
,选项C对。用能量为10.3 eV的光子照射,由于该光子能量不等于任意两个氢原子的能极差,而且小于从基态到电离所需的能量,所以基态的氢原子不能吸收该光子跃迁,而14.0 eV的光子足以使得氢原子从基态电离,多余的能量转化为电离后的动能,选项D错E对。
练习册系列答案
相关题目






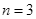 的能级跃迁到
的能级跃迁到 的能级,该氢原子吸收光子,能量增加
的能级,该氢原子吸收光子,能量增加  )经过多次
)经过多次 、
、 衰变形成稳定的铅(
衰变形成稳定的铅(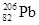 )的过程中,有6个中子转变成质子
)的过程中,有6个中子转变成质子 射线较强的穿透能力
射线较强的穿透能力